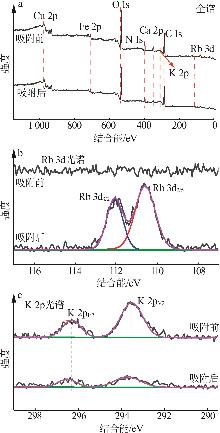
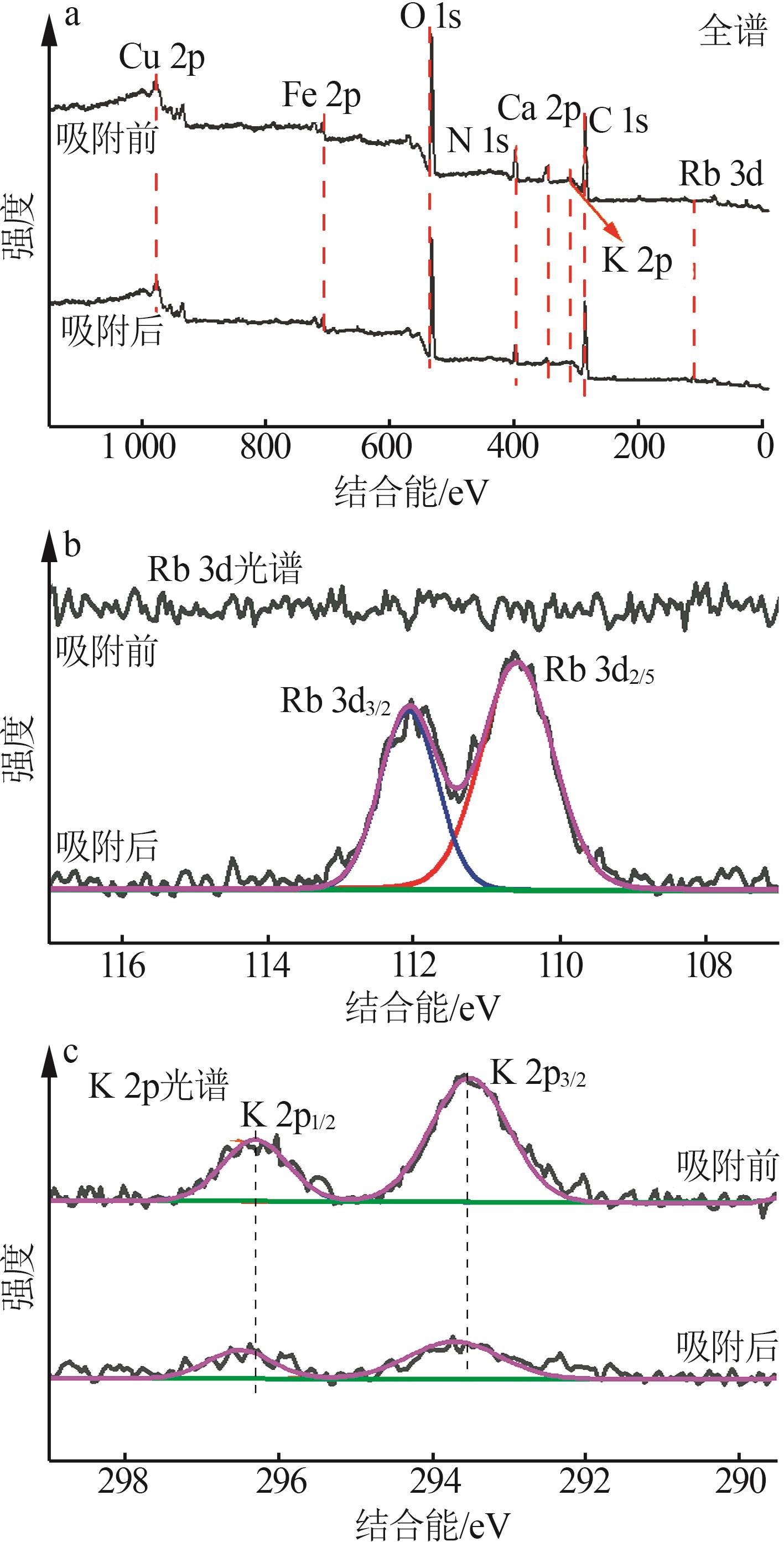
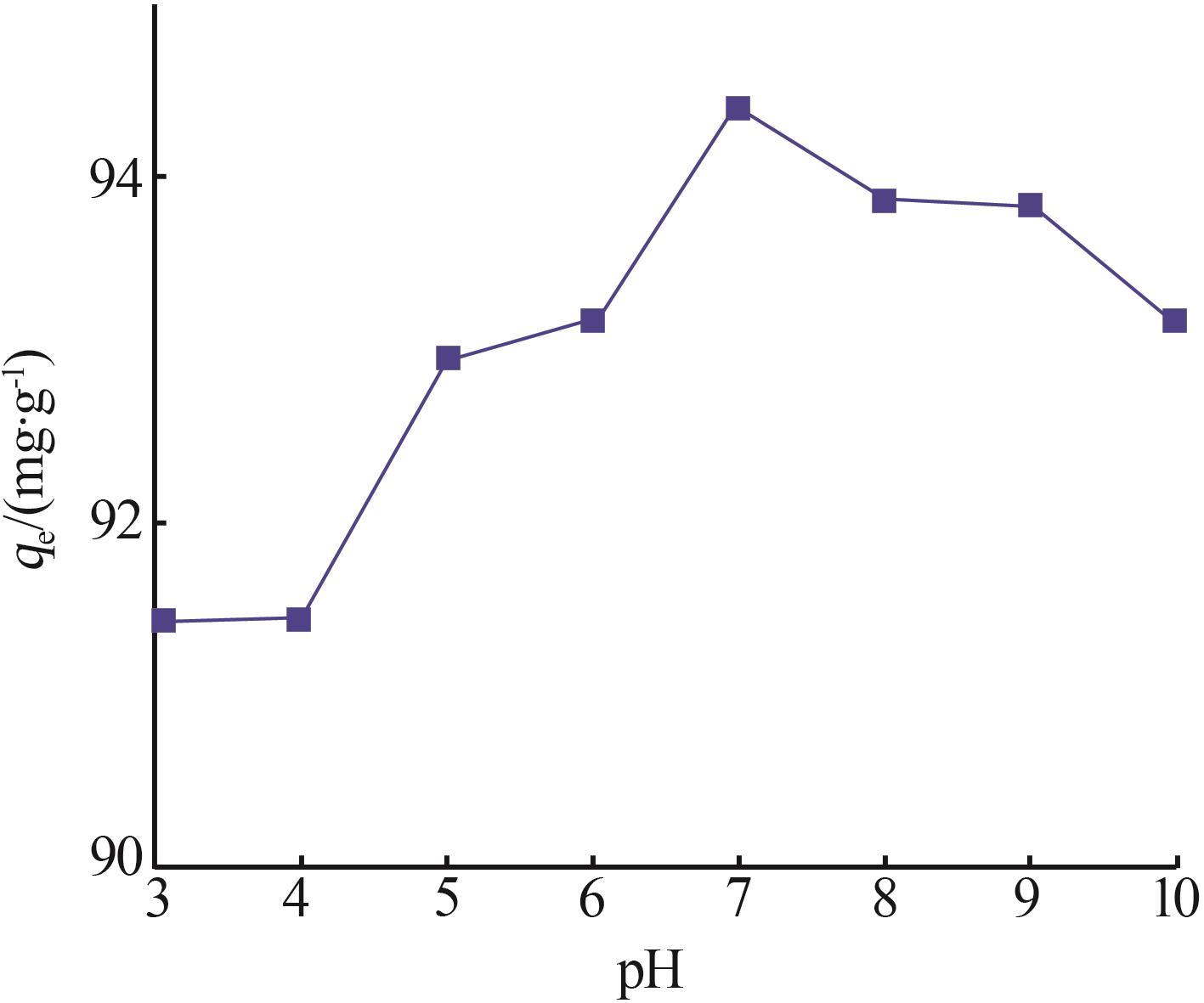
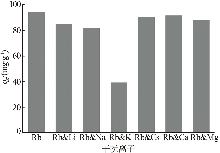
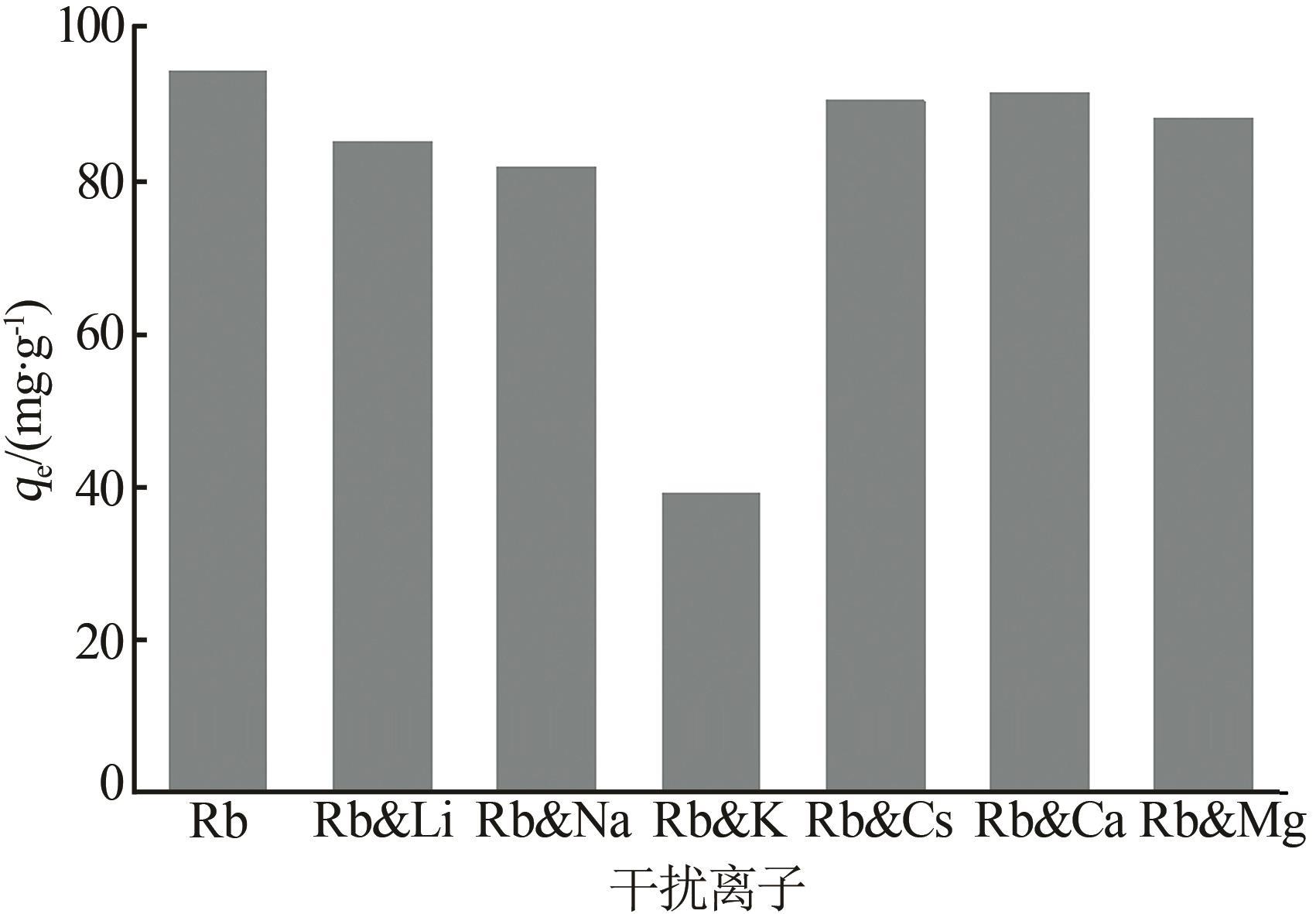
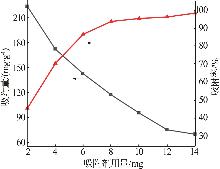
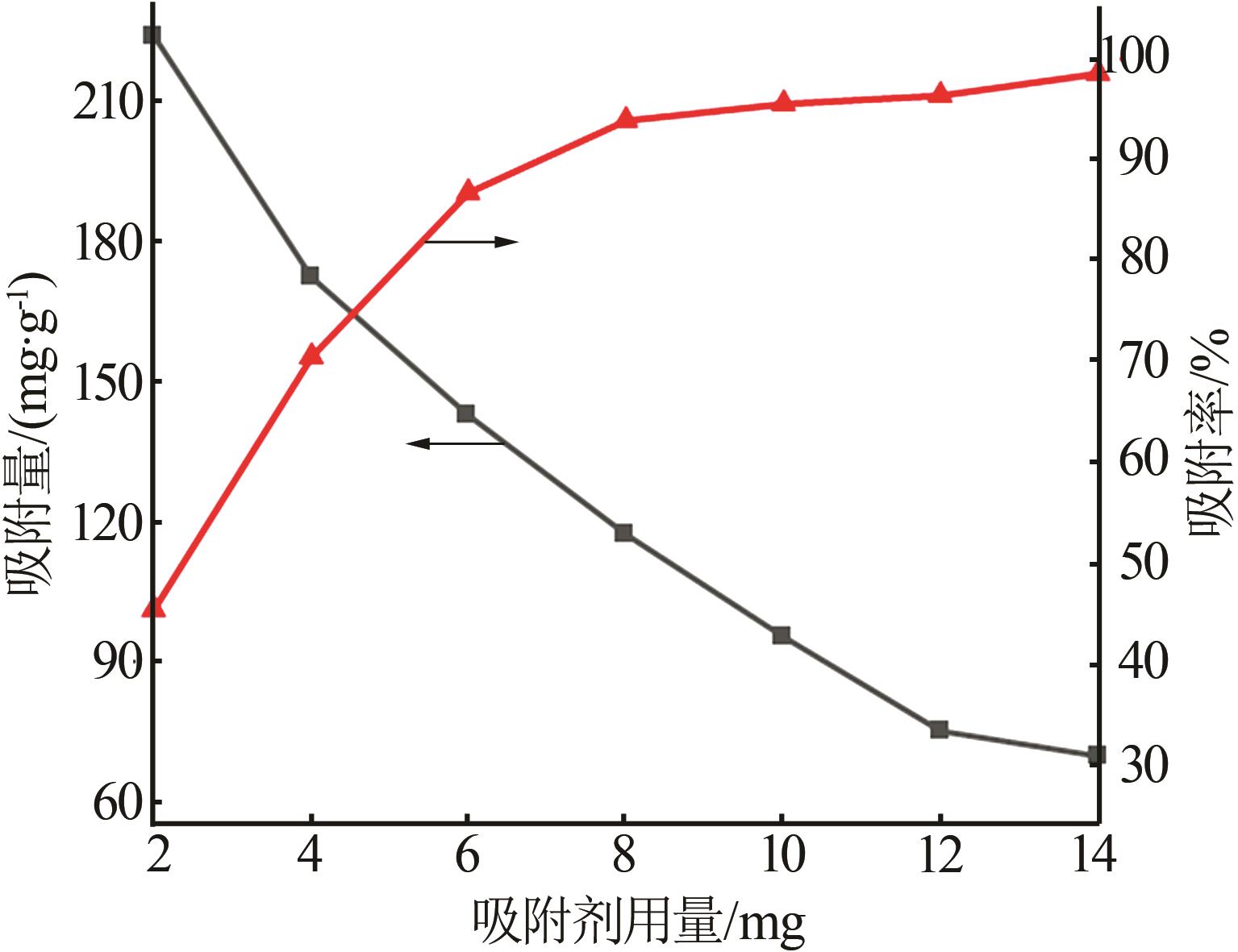
Inorganic Chemicals Industry ›› 2026, Vol. 58 ›› Issue (3): 32-40.doi: 10.19964/j.issn.1006-4990.2025-0100
• Research & Development • Previous Articles Next Articles
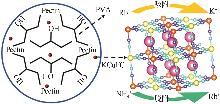
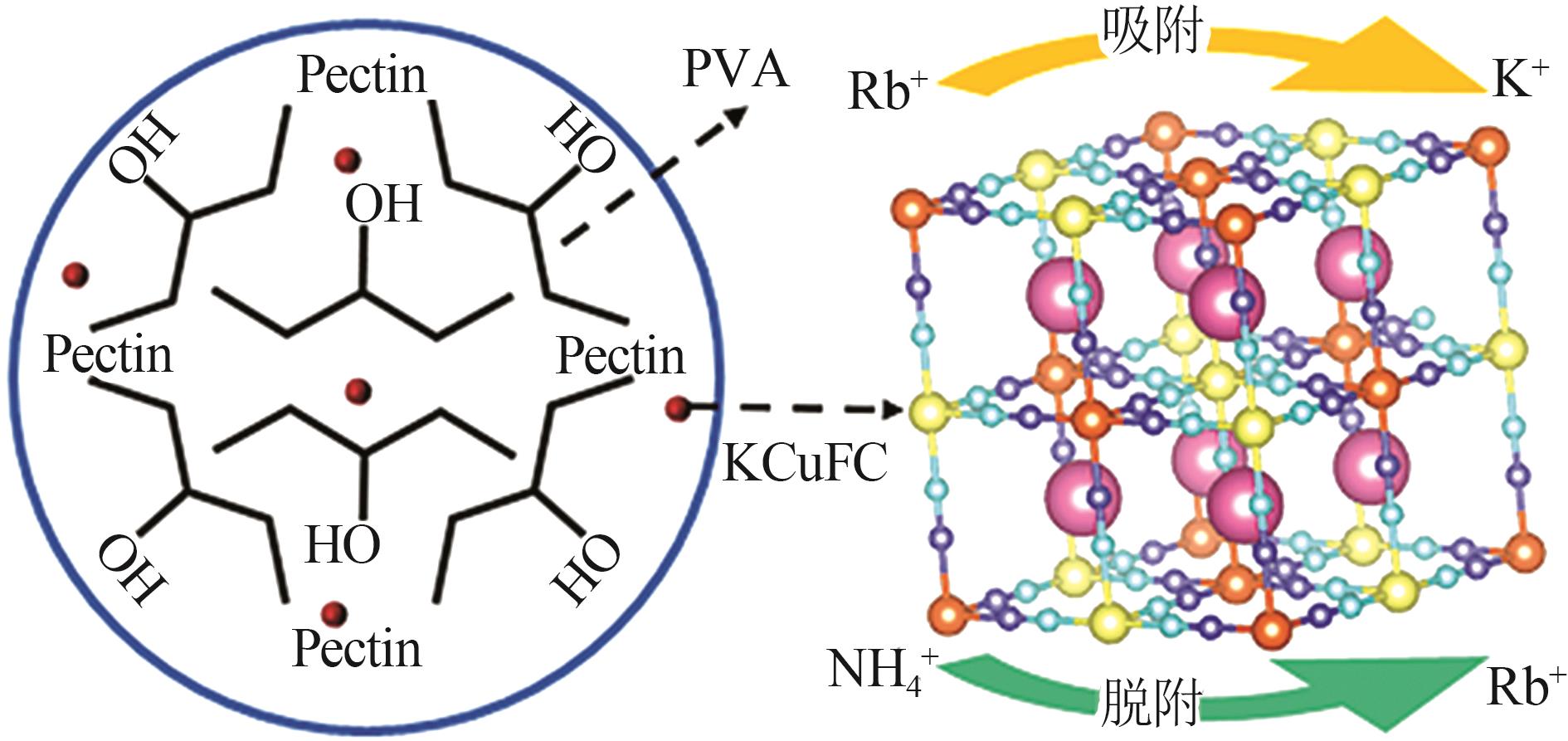
Study on preparation of potassium copper ferrocyanide composite hydrogels and rubidium adsorption
LIN Peihua( ), JIANG Huaitao, FU Yibo
), JIANG Huaitao, FU Yibo
- College of Chemistry and Chemical Engineering,Hefei University of Technology,Hefei 230009,China
-
Received:2025-02-26Online:2026-03-10Published:2025-08-12
CLC Number:
Cite this article
LIN Peihua, JIANG Huaitao, FU Yibo. Study on preparation of potassium copper ferrocyanide composite hydrogels and rubidium adsorption[J]. Inorganic Chemicals Industry, 2026, 58(3): 32-40.
share this article
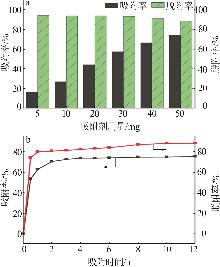
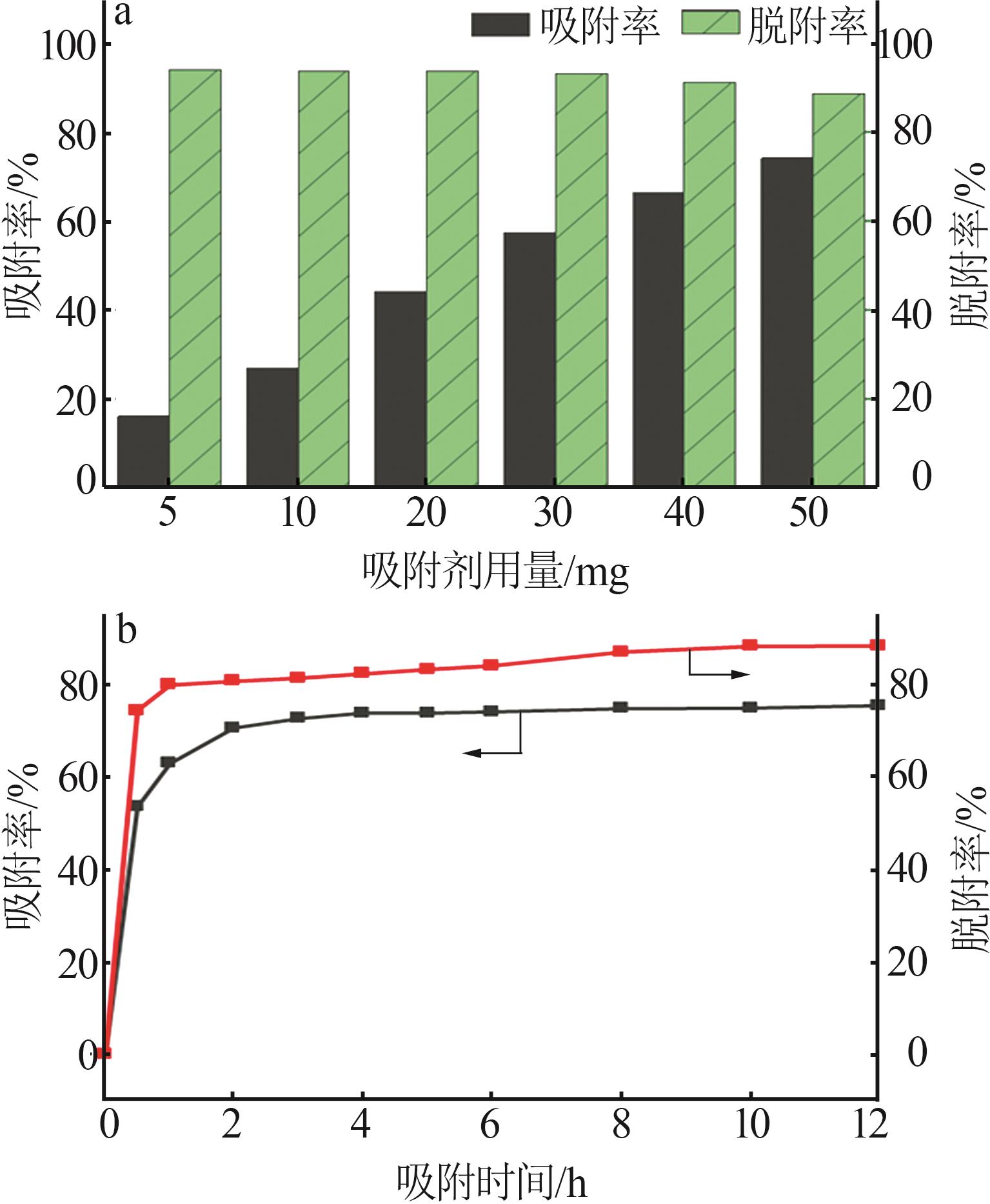
Table 4
Mass concentrations of various metal ions in raw brine and after adsorption with different adsorbent dosages"
| 吸附剂用量/mg | ρ(Li+)/(mg·L-1) | ρ(Na+)/(mg·L-1) | ρ(Mg2+)/(mg·L-1) | ρ(K+)/ (mg·L-1) | ρ(Rb+)/(mg·L-1) | ρ(Cs+)/(mg·L-1) |
|---|---|---|---|---|---|---|
| 0 | 7.29 | 1 139.65 | 10 241.57 | 2 655.88 | 9.87 | 0.14 |
| 5 | 7.21 | 1 137.12 | 10 148.24 | 2 667.90 | 8.27 | 0.02 |
| 10 | 7.16 | 1 106.41 | 10 022.20 | 2 674.43 | 7.20 | — |
| 20 | 7.15 | 1 081.55 | 9 777.37 | 2 711.11 | 5.49 | — |
| 30 | 7.08 | 1 076.87 | 9 933.63 | 2 737.79 | 4.18 | — |
| 40 | 7.00 | 1 072.18 | 9 719.92 | 2 791.01 | 3.31 | — |
| 50 | 6.99 | 1 066.28 | 9 819.51 | 2 807.13 | 2.53 | — |
Table 5
Mass concentration of various metal ions in desorption solution"
吸附剂 用量/mg | ρ(Li+)/(mg·L-1) | ρ(Na+)/(mg·L-1) | ρ(Mg2+)/(mg·L-1) | ρ(K+)/(mg·L-1) | ρ(Rb+)/(mg·L-1) | ρ(Cs+)/(mg·L-1) |
|---|---|---|---|---|---|---|
| 5 | 0.22 | — | 30.97 | 4.30 | 1.51 | — |
| 10 | 0.22 | — | 62.69 | 13.82 | 2.51 | — |
| 20 | 0.25 | — | 128.28 | 30.80 | 4.11 | — |
| 30 | 0.25 | — | 157.67 | 35.65 | 5.32 | — |
| 40 | 0.28 | 0.12 | 229.12 | 53.75 | 6.00 | — |
| 50 | 0.35 | 1.15 | 295.56 | 69.25 | 6.52 | — |
| [1] | XING Peng, WANG Chengyan, CHEN Yongqiang,et al.Rubidium extraction from mineral and brine resources:A review[J].Hydro- metallurgy,2021,203:105644. |
| [2] | LV Yingwei, XING Peng, MA Baozhong,et al.Efficient extraction of lithium and rubidium from polylithionite via alkaline leaching combined with solvent extraction and precipitation[J].ACS Sustainable Chemistry & Engineering,2020,8(38):14462-14470. |
| [3] | WANG Ling, XIONG Yanhang, ZHOU Xiaowei,et al.Complexation mechanism of crown ethers with rubidium and cesium ions using density functional theory[J].Computational and Theoretical Chemistry,2023,1225:114139. |
| [4] | LIU Haining, WANG Yanping, ZHANG Qiongyuan,et al.High-efficiency selective adsorption of rubidium and cesium from simulated brine using a magnesium ammonium phosphate adsorbent[J].Separations,2024,11(9):277. |
| [5] | SHI Qiang, ZHANG Haiyan, JIAO Jixuan,et al.Poly(styrene-co-4-hydroxystyrene) nanofiber membrane for highly selective and efficient Rb+ capture from high salinity solution[J].Separation and Purification Technology,2023,312:123334. |
| [6] | NAIDU G, LOGANATHAN P, JEONG S,et al.Rubidium extraction using an organic polymer encapsulated potassium copper he-xacyanoferrate sorbent[J].Chemical Engineering Journal,2016,306:31-42. |
| [7] | 柳富杰,何倩,苏龙,等.海藻酸钠表面功能化磁性生物炭对亚甲基蓝的吸附特性[J].无机盐工业,2024,56(2):65-73. |
| LIU Fujie, HE Qian, SU Long,et al.Adsorption properties of methylene blue by surface functionalized magnetic biochar with sodium alginate[J].Inorganic Chemicals Industry,2024,56(2):65-73. | |
| [8] | YANG Xiaobo, LI Wei, LIN Zezhong,et al.Ultralight,robust,and high Prussian blue-loading polyacrylonitrile aerogel:Preparation,characterization and efficient adsorption/removal of Cs+ [J].Chemical Engineering Journal,2023,464:142723. |
| [9] | FANG Ming, CHU Weifan, CUI Jingsi,et al.Adsorption application of Rb+ on hydrogels of hydroxypropyl cellulose/polyvinyl alcohol/reduced graphene oxide encapsulating potassium cobalt hexacyanoferrate[J].Applied Organometallic Chemistry,2022,36(10):e6827. |
| [10] | YOON S, CHOI M, HWANG Y,et al.Upcycling of steel slag for manufacture of Prussian-blue-encapsulated pectin beads and its use for efficient removal of aqueous cesium[J].Journal of Cleaner Production,2021,319:128786. |
| [11] | KHIÊM V N, MAI T T, URAYAMA K,et al.A multiaxial theory of double network hydrogels[J].Macromolecules,2019,52(15):5937-5947. |
| [12] | WANG Yun, LI Heng, LI Jinming,et al.Porous PVA-PAM-AG multi-network hydrogel with high mechanical strength[J].Journal of Applied Polymer Science,2025,142(18):e56836. |
| [13] | 胡冬婉,马占玲,马骁,等.改性果胶-Fe3O4磁性微球制备及对Pb2+吸附性能[J].无机盐工业,2020,52(6):24-29. |
| HU Dongwan, MA Zhanling, MA Xiao,et al.Preparation of modified Fe3O4-pectin magnetic microspheres and its adsorption property for Pb2+ [J].Inorganic Chemicals Industry,2020,52(6):24-29. | |
| [14] | 许梦丽,王浩,刘志华,等.聚乙烯醇与硼酸改性醋酸纤维素的性能[J].功能高分子学报,2024,37(6):509-515. |
| XU Mengli, WANG Hao, LIU Zhihua,et al.Properties of cellulose acetate modified with polyvinyl alcohol and boric acid[J].Journal of Functional Polymers,2024,37(6):509-515. | |
| [15] | ABDUL WAHAB M S, RAHMAN S A, SAMAH R ABU.Hydrophilic enhancement of polysulfone membrane via graphene oxide embedded thin film nanocomposite for Isopropanol dehydrat- ion[J].Vacuum,2020,180:109569. |
| [16] | CHU Weifan, YUAN Zhouzhou, LIN Peihua,et al.Recovery of rubidium using hydrogel beads encapsulating potassium copper hexacyanoferrate from saline lake brines[J].Industrial & Engineering Chemistry Research,2024,63(4):1988-1999. |
| [17] | LUO Liming, PENG Tongjiang, YUAN Mingliang,et al.Preparation of graphite oxide containing different oxygen-containing functional groups and the study of ammonia gas sensitivity[J].Sensors,2018,18(11):3745. |
| [18] | MARY I A, SELVANAYAGAM S, SELVASEKARAPANDIAN S,et al.Lithium ion conducting biopolymer membrane based on kappa carrageenan with LiCl and its application to electrochemical devices[J].Materials Today:Proceedings,2022,58:855-861. |
| [19] | 彭良琼,林诗雨,郭丽君,等.含铬革屑负载亚铁氰化铜钾对铯的快速吸附[J].皮革科学与工程,2023,33(5):8-15. |
| PENG Liangqiong, LIN Shiyu, GUO Lijun,et al.Rapid adsorption of cesium by potassium copper ferrocyanide loaded on chro-me shavings[J].Leather Science and Engineering,2023,33(5):8-15. | |
| [20] | ABURTO J, MORAN M, GALANO A,et al.Non-isothermal pyrolysis of pectin:A thermochemical and kinetic approach[J].Journal of Analytical and Applied Pyrolysis,2015,112:94-104. |
| [21] | WANG Qianqian, HE Sifa, WANG Ning,et al.Synthesis of CoFe Prussian blue analogue/carbon nanotube composite material and its application in the catalytic epoxidation of styrene[J].New Jo- urnal of Chemistry,2016,40(4):3244-3251. |
| [22] | ÁVILA-ORTA C A, SORIANO CORRAL F, FONSECA-FLORIDO H A,et al.Starch-graphene oxide bionanocomposites prepared through melt mixing[J].Journal of Applied Polymer Science,2018,135(12):46037. |
| [23] | ZHANG Lihong, LI Yun, LIN Nana,et al.Significantly enhanced alkaline stability and cyanide suppression of Prussian blue analogues using montmorillonite for high-performance cesium remo-val[J].Separation and Purification Technology,2023,325:124662. |
| [24] | BOK-BADURA J, KAZEK-KĘSIK A, KAROŃ K,et al.Highly efficient copper hexacyanoferrate-embedded pectin sorbent for radioactive cesium ions removal[J].Water Resources and Industry,2022,28:100190. |
| [25] | YI Rong, YE Gang, WU Fengcheng,et al.Highly efficient removal of 137Cs in seawater by potassium titanium ferrocyanide functionalized magnetic microspheres with multilayer core-shell structure[J].RSC Advances,2014,4(71):37600-37608. |
| [26] | DURANOĞLU D, TROCHIMCZUK A W, BEKER U.Kinetics and thermodynamics of hexavalent chromium adsorption onto activated carbon derived from acrylonitrile-divinylbenzene copolymer[J].Chemical Engineering Journal,2012,187:193-202. |
| [27] | WANG Jianlong, GUO Xuan.Adsorption isotherm models:Classification,physical meaning,application and solving method[J].Chemosphere,2020,258:127279. |
| [1] | XU Fan, YIN Xiaoyan, SONG Feng. Study on low⁃temperature synthesis of low⁃silica CHA zeolite and its CO2/N2 adsorption and separation performance [J]. Inorganic Chemicals Industry, 2026, 58(3): 41-48. |
| [2] | LI Lei, HAN Enshan, LI Bin, SUN Zhenhai, HE Yanzhen. Study on microwave modification of activated carbon by ammonia and its adsorption for separation of polycyclic aromatic hydrocarbons [J]. Inorganic Chemicals Industry, 2026, 58(3): 65-71. |
| [3] | WU Jie, XU Chunhui, WANG Feng, TANG Zhongfeng. Effect of NaNO3 doping on structure and adsorption-desorption performance of MgO-based CO2 adsorbents [J]. Inorganic Chemicals Industry, 2026, 58(2): 60-67. |
| [4] | HONG Menghuan, NASEN Bate, WANG Yaya, LI Shuangqin, XU Dehua, YAN Zhengjuan, LIU Wei, WANG Xinlong. Study on preparation of biochar via pyrolysis of acid-base synergistically treated chicken manure and its adsorption performance for Cd²⁺ [J]. Inorganic Chemicals Industry, 2026, 58(2): 68-75. |
| [5] | WU Fenglong, SONG Jin. Study on photocatalytic synthesis of low molecular weight anti⁃mud polycarboxylate superplasticizers and its adsorption effect for kaolin [J]. Inorganic Chemicals Industry, 2026, 58(1): 52-60. |
| [6] | WANG Guibin, ZHANG Hongwei, LIU Lijuan, ZHAO Yu, ZHANG Dongqiang. Study on effect of resin modification on removal performance of fluoride and chloride ions in desulfurization ionic liquid [J]. Inorganic Chemicals Industry, 2026, 58(1): 18-25. |
| [7] | CHEN Jianjun, LI Li, LIU Laibao, ZHANG Daiyu, JIANG Chenxi, ZHU Hanzhen, WANG Fu, LIAO Qilong. Study on synthesis of high specific surface area calcium hydroxide by quicklime digestion and its adsorption performance for lead [J]. Inorganic Chemicals Industry, 2026, 58(1): 99-107. |
| [8] | CHEN Mengmeng, XU Dekan, HUANG Jilong, TANG Zhilan, ZHANG Xu, TAN Chao, WANG Xiaohu, PENG Wenbo. Study on preparation and performance of Ni-modified titanium-based lithium ion sieve [J]. Inorganic Chemicals Industry, 2025, 57(9): 37-45. |
| [9] | WANG Zhigang, HU Xiaodong, WANG Hui, XUE Lianggang, TIAN Jianqiang. Study on preparation of NO2- modified hydrotalcite and its corrosion resistance to steel bars [J]. Inorganic Chemicals Industry, 2025, 57(8): 74-81. |
| [10] | HAN Xing, SHI Dandan, WANG Xingquan, ZHANG Tianying, CAO Yue, WANG Yiying, ZHU Xiang, XU Naicai. Study on Mg-Al hydrotalcite modified by sodium dodecyl sulfate and its adsorption performance for Pb2+ [J]. Inorganic Chemicals Industry, 2025, 57(8): 48-57. |
| [11] | ZHAO Xueqing, WANG Zhonghui, WANG Yongwei, WANG Zhiyu, HUO Xiaodong. Study on preparation of nitrogen-doped porous carbon materials derived from used cigarette filters and their adsorption properties [J]. Inorganic Chemicals Industry, 2025, 57(7): 110-119. |
| [12] | YANG Hengyu, MO Hengliang, LI Tianyu, LÜ Long, CHEN Yili, WANG Luocong, ZHAO Wenfang, LIU Manman. Study on synthesis of high acid resistant Ti-based lithium ion sieve using mixed crystal TiO2 as titanium source [J]. Inorganic Chemicals Industry, 2025, 57(7): 57-63. |
| [13] | ZHANG Hanfei, SHEN Hongyan, LIU Youzhi. Study on preparation of MG composite material and its adsorption properties for methyl orange [J]. Inorganic Chemicals Industry, 2025, 57(6): 100-107. |
| [14] | PANG Hao, LI Bin, SUN Zhenhai, GUO Jian, CAI Zhe, WU Luming, HONG Meihua, YANG Zhanfeng. Study on adsorption and denitrification performance of iron doped modified SiO2 [J]. Inorganic Chemicals Industry, 2025, 57(6): 49-55. |
| [15] | SUN Mengyuan, HE Wei, WANG Chen. Research progress of preparation and adsorption of magnesium oxide fibers and their composite fibers [J]. Inorganic Chemicals Industry, 2025, 57(5): 1-10. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||
|
||