Inorganic Chemicals Industry ›› 2026, Vol. 58 ›› Issue (2): 60-67.doi: 10.19964/j.issn.1006-4990.2024-0655
• Research & Development • Previous Articles Next Articles
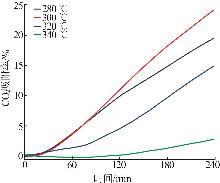
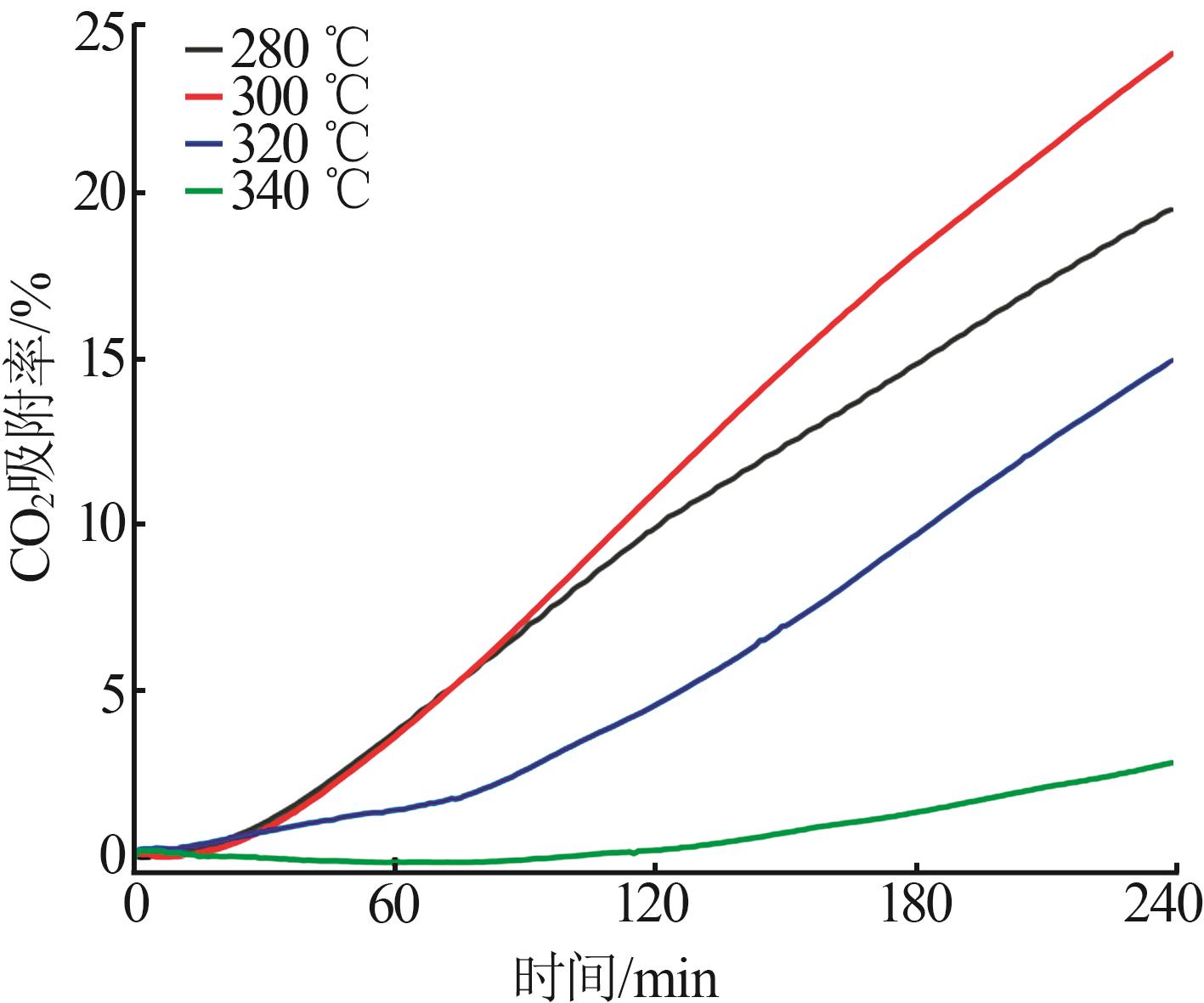
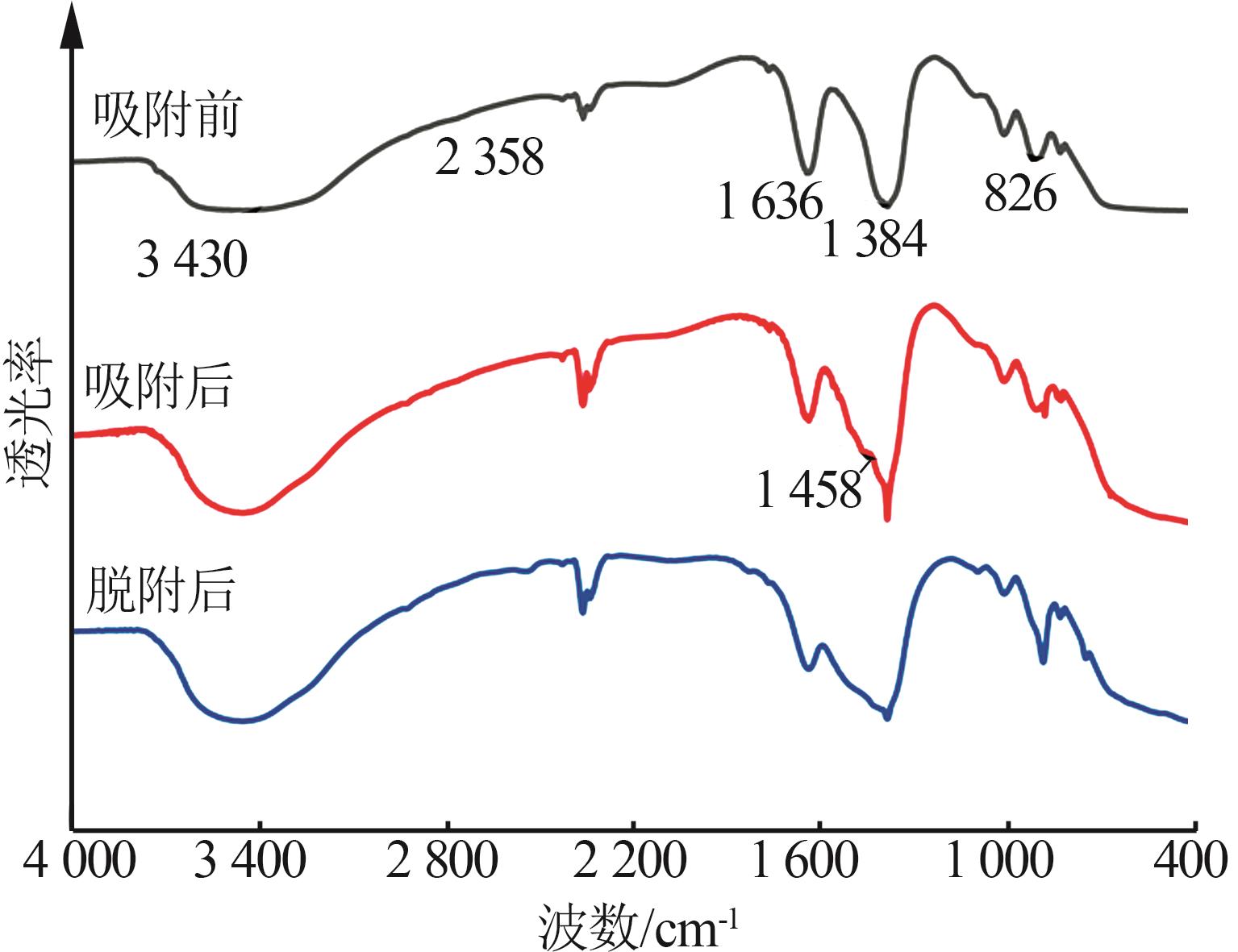
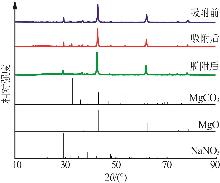
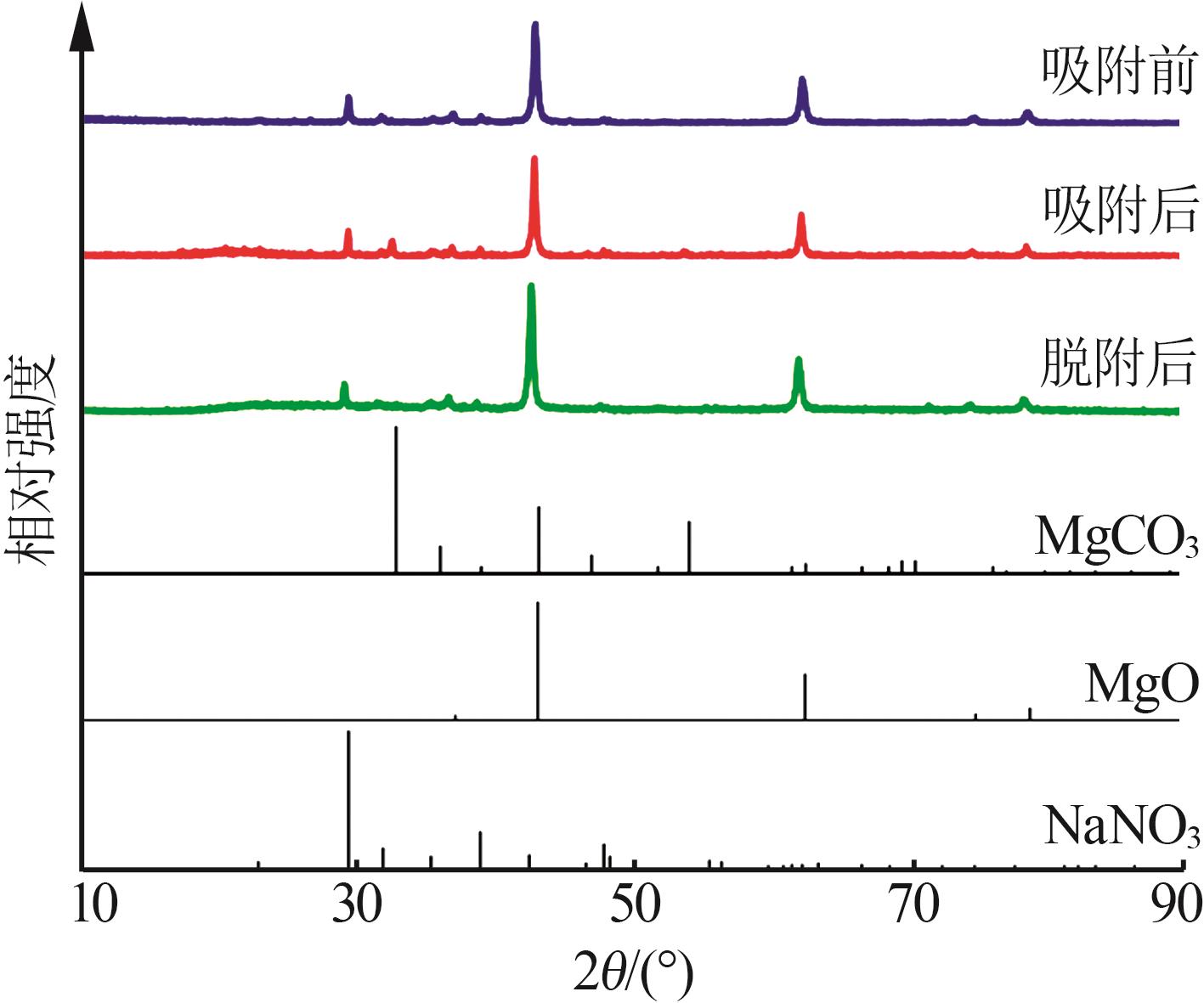
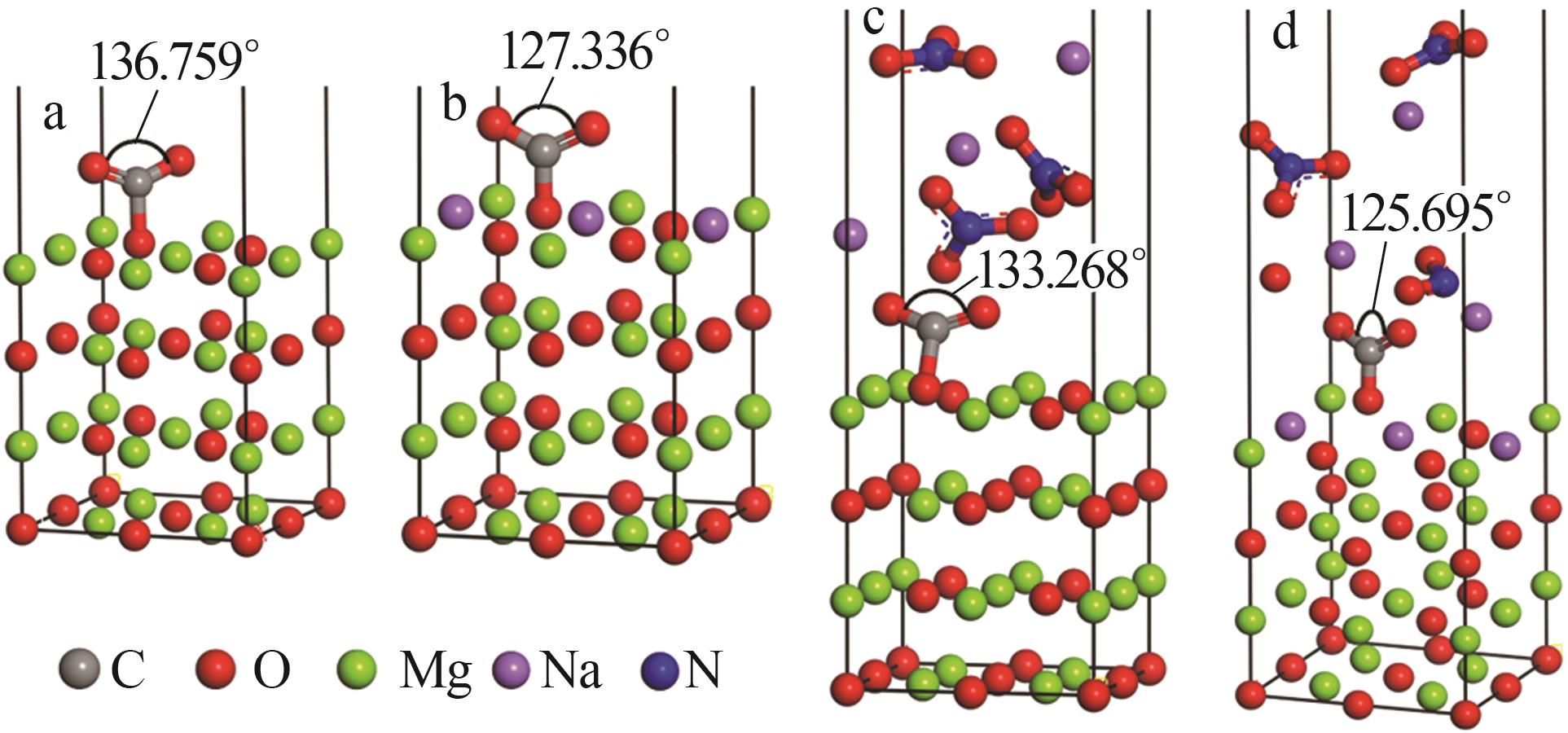
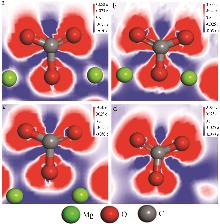
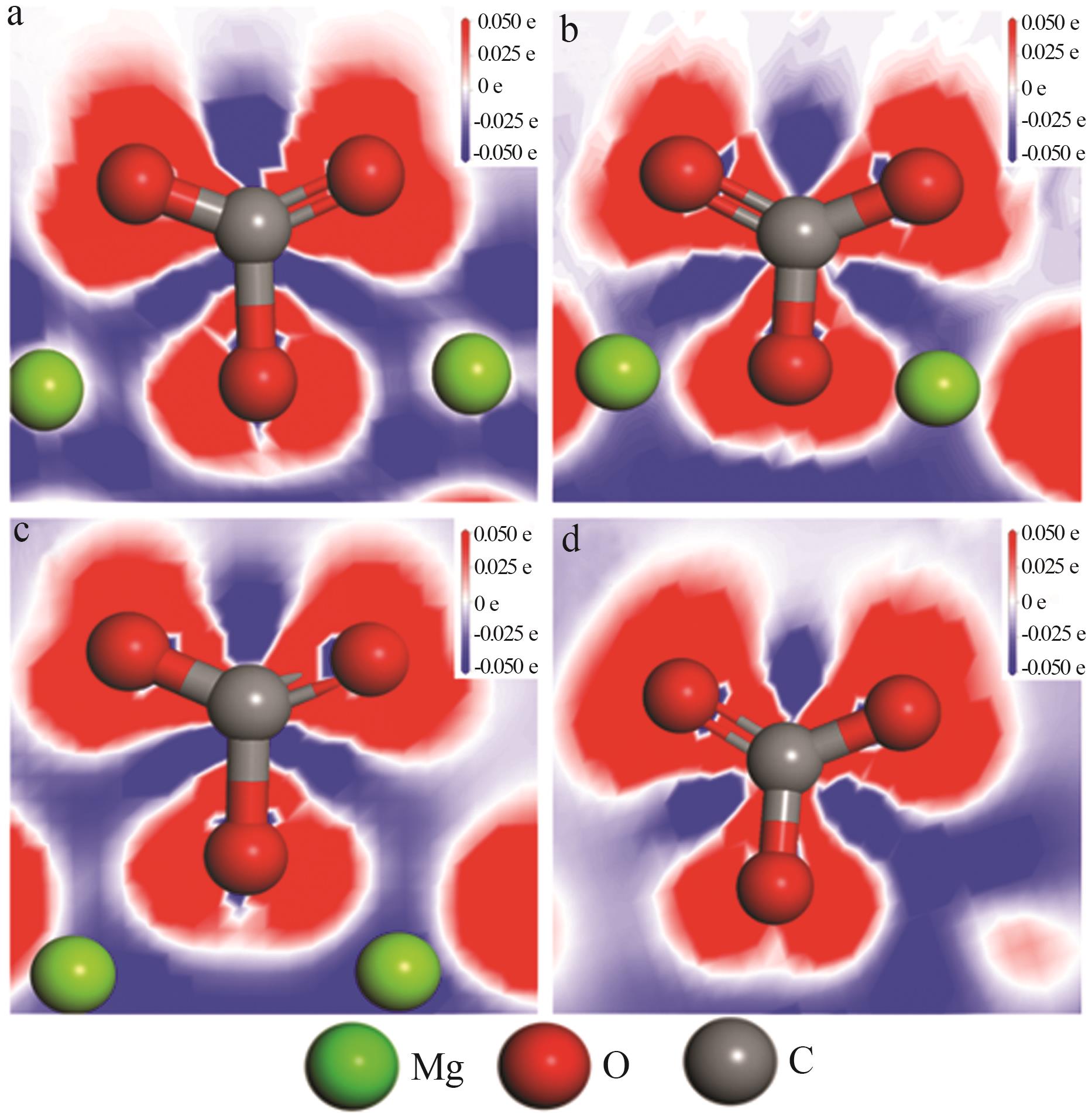
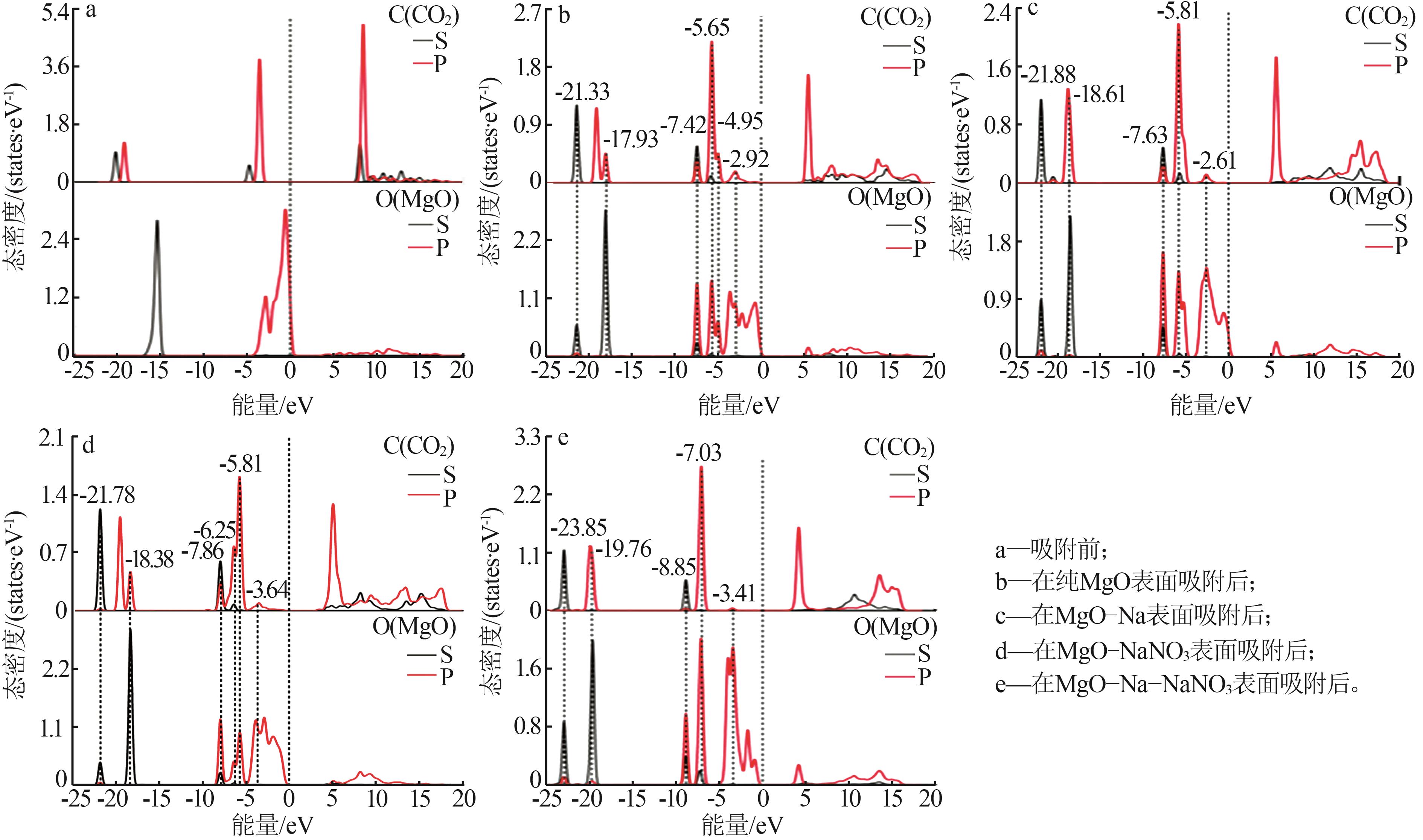
Effect of NaNO3 doping on structure and adsorption-desorption performance of MgO-based CO2 adsorbents
WU Jie1,2( ), XU Chunhui2, WANG Feng2(
), XU Chunhui2, WANG Feng2( ), TANG Zhongfeng3(
), TANG Zhongfeng3( )
)
- 1.Inner Mongolia Power Research Institute Branch,Inner Mongolia Power(Group) Co. ,Ltd. ,Hohhot 010020,China
2.School of Energy and Power Engineering,Inner Mongolia University of Technology,Hohhot 010051,China
3.Shanghai Institute of Applied Physics,Chinese Academy of Sciences,Shanghai 201800,China
-
Received:2025-12-04Online:2026-02-10Published:2025-08-01 -
Contact:WANG Feng, TANG Zhongfeng E-mail:wujiegongda@126.com;wangfeng@imut.com;tangzhongfeng@sinap.ac.cn
CLC Number:
Cite this article
WU Jie, XU Chunhui, WANG Feng, TANG Zhongfeng. Effect of NaNO3 doping on structure and adsorption-desorption performance of MgO-based CO2 adsorbents[J]. Inorganic Chemicals Industry, 2026, 58(2): 60-67.
share this article
| [1] | 许春辉,王峰,凌长见,等.熔盐改性的金属氧化物捕获二氧化碳研究进展[J].无机盐工业,2023,55(5):1-7,23. |
| XU Chunhui, WANG Feng, LING Changjian,et al.Research progress of CO2 capture by metal oxides modified by molten salts[J].Inorganic Salt Industry,2023,55(5):1-7,23. | |
| [2] | 桂昌青,王雅静,凌长见,等.氧化镁基二氧化碳吸附剂的制备及改性研究进展[J].无机盐工业,2023,55(8):77-83. |
| GUI Changqing, WANG Yajing, LING Changjian,et al.Research progress of preparation and modification of MgO-based CO2 adsorbents[J].Inorganic Chemicals Industry,2023,55(8):77-83. | |
| [3] | LIU Song, CHENG Guojun, TANG Zhongfeng.Effect of succinic acid on morphology transformation of the anhydrous magnesium carbonate particles prepared by magnesium chloride hexahydrate and urea[J].Particuology,2024,90:209-217. |
| [4] | LI Ping, LIU Bingxin, LAI Xing,et al.Thermal decomposition mechanism and pyrolysis products of waste bischofite calcined at high temperature[J].Thermochimica Acta,2022,710:179164. |
| [5] | GÜR T M.Carbon dioxide emissions,capture,storage and utilization:Review of materials,processes and technologies[J].Progress in Energy and Combustion Science,2022,89:100965. |
| [6] | POZZO A DAL, ARMUTLULU A, REKHTINA M,et al.CO2 uptake and cyclic stability of MgO-based CO2 sorbents promoted with alkali metal nitrates and their eutectic mixtures[J].ACS Applied Energy Materials,2019,2(2):1295-1307. |
| [7] | XU Chunhui, WANG Zirui, WANG Feng,et al.NaNO3-promoted MgO-based adsorbents prepared from bischofite for CO2 capture:Experimental and density functional theory study[J].Langmuir,2024,40(9):5001-5010. |
| [8] | GUI Changqing, WANG Zirui, LING Changjian,et al.Molten salt-promoted MgO-based CO2 adsorbents:Selective adsorption on polycrystalline surfaces[J].Journal of Environmental Chemical Engineering,2024,12(2):111972. |
| [9] | HARADA T, SIMEON F, HAMAD E Z,et al.Alkali metal nitrate-promoted high-capacity MgO adsorbents for regenerable CO2 capture at moderate temperatures[J].Chemistry of Materials,2015,27(6):1943-1949. |
| [10] | PARK S J, KIM Y, JONES C W.NaNO3-promoted mesoporous MgO for high-capacity CO2 capture from simulated flue gas with isothermal regeneration[J].ChemSusChem,2020,13(11):2988-2995. |
| [11] | QIAO Yaqian, WANG Junya, ZHANG Yu,et al.Alkali nitrates molten salt modified commercial MgO for intermediate-temperature CO2 capture:Optimization of the Li/Na/K ratio[J].Industrial & Engineering Chemistry Research,2017,56(6):1509-1517. |
| [12] | PANG Hua, SUN Anwei, XU Haoran,et al.Regenerable MgO-based sorbents for CO2 capture at elevated temperature and pressure:Experimental and DFT study[J].Chemical Engineering Journal,2021,425:130675. |
| [13] | GAO Wanlin, XIAO Jiewen, WANG Qiang,et al.Unravelling the mechanism of intermediate-temperature CO2 interaction with mo-lten-NaNO3-salt-promoted MgO[J].Advanced Materials,2022,34(4):2106677. |
| [14] | LANDUYT A, KUMAR P V, YUWONO J A,et al.Uncovering the CO2 capture mechanism of NaNO3-promoted MgO by 18O isotope labeling[J].JACS Au,2022,2(12):2731-2741. |
| [15] | GAO Wanlin, VASILIADES M A, DAMASKINOS C M,et al.Molten salt-promoted MgO adsorbents for CO2 capture:Transient kinetic studies[J].Environmental Science & Technology,2021,55(8):4513-4521. |
| [16] | BORK A H, REKHTINA M, WILLINGER E,et al.Peering into buried interfaces with X-rays and electrons to unveil MgCO3 formation during CO2 capture in molten salt-promoted MgO[J].Proceedings of the National Academy of Sciences of the United States of America,2021,118(26):e2103971118. |
| [17] | 徐云轩,林尚超,闫君,等.MgCO3/MgO热化学储热的碳酸化反应动力学机理研究[J].工程热物理学报,2023,44(2):468-474. |
| XU Yunxuan, LIN Shangchao, YAN Jun,et al.Kinetic mechanism of carbonation reaction of MgCO3/MgO thermochemical energy storage[J].Journal of Engineering Thermophysics,2023,44(2):468-474. | |
| [18] | KWON S, CHOI J I, LEE S G,et al.A density functional theory (DFT) study of CO2 adsorption on Mg-rich minerals by enhanc- |
| ed charge distribution[J].Computational Materials Science,2014,95:181-186. | |
| [19] | ZHOU Siyu, ZHOU Yan, LING Ziye,et al.Modification of expanded graphite and its adsorption for hydrated salt to prepare composite PCMs[J].Applied Thermal Engineering,2018,133:446-451. |
| [20] | CUI Ruizhi, LI Wu, DONG Yaping,et al.Mineral solubilities of salts in the three quaternary systems:LiCl-NaCl-MgCl2-H2O,LiCl-KCl-MgCl2-H2O and Li2SO4-K2SO4-MgSO4-H2O at 288.15 K[J].The Journal of Chemical Thermodynamics,2019,138:127-139. |
| [21] | 马广超,狄跃忠,彭建平,等.青海盐湖水氯镁石利用技术现状[J].矿产保护与利用,2019,39(3):160-166. |
| MA Guangchao, DI Yuezhong, PENG Jianping,et al.Utilization technical status of bischofite in Qinghai salt lake[J].Conservation and Utilization of Mineral Resources,2019,39(3):160-166. | |
| [22] | ZHANG Keling, LI X S, LI Weizhen,et al.Phase transfer-catalyzed fast CO2 absorption by MgO-based absorbents with high cycling capacity[J].Advanced Materials Interfaces,2014,1(3):1400030. |
| [23] | DING Jing, YU Chao, LU Jianfeng,et al.Enhanced CO2 adsorption of MgO with alkali metal nitrates and carbonates[J].Applied Energy,2020,263:114681. |
| [1] | HONG Menghuan, NASEN Bate, WANG Yaya, LI Shuangqin, XU Dehua, YAN Zhengjuan, LIU Wei, WANG Xinlong. Study on preparation of biochar via pyrolysis of acid-base synergistically treated chicken manure and its adsorption performance for Cd²⁺ [J]. Inorganic Chemicals Industry, 2026, 58(2): 68-75. |
| [2] | WU Fenglong, SONG Jin. Study on photocatalytic synthesis of low molecular weight anti⁃mud polycarboxylate superplasticizers and its adsorption effect for kaolin [J]. Inorganic Chemicals Industry, 2026, 58(1): 52-60. |
| [3] | WANG Guibin, ZHANG Hongwei, LIU Lijuan, ZHAO Yu, ZHANG Dongqiang. Study on effect of resin modification on removal performance of fluoride and chloride ions in desulfurization ionic liquid [J]. Inorganic Chemicals Industry, 2026, 58(1): 18-25. |
| [4] | CHEN Jianjun, LI Li, LIU Laibao, ZHANG Daiyu, JIANG Chenxi, ZHU Hanzhen, WANG Fu, LIAO Qilong. Study on synthesis of high specific surface area calcium hydroxide by quicklime digestion and its adsorption performance for lead [J]. Inorganic Chemicals Industry, 2026, 58(1): 99-107. |
| [5] | ZHANG Longhua, ZHANG Zhichao. Study on synergistic modification to improve structural stability of ternary cathode material LiNi0.9Co0.05Mn0.05O2 [J]. Inorganic Chemicals Industry, 2025, 57(9): 82-87. |
| [6] | CHEN Mengmeng, XU Dekan, HUANG Jilong, TANG Zhilan, ZHANG Xu, TAN Chao, WANG Xiaohu, PENG Wenbo. Study on preparation and performance of Ni-modified titanium-based lithium ion sieve [J]. Inorganic Chemicals Industry, 2025, 57(9): 37-45. |
| [7] | SHEN Mengmeng. Study on preparation of g-C3N4/polyaniline composites and their application in photocatalytic reduction of CO2 [J]. Inorganic Chemicals Industry, 2025, 57(8): 123-130. |
| [8] | WANG Zhigang, HU Xiaodong, WANG Hui, XUE Lianggang, TIAN Jianqiang. Study on preparation of NO2- modified hydrotalcite and its corrosion resistance to steel bars [J]. Inorganic Chemicals Industry, 2025, 57(8): 74-81. |
| [9] | HAN Xing, SHI Dandan, WANG Xingquan, ZHANG Tianying, CAO Yue, WANG Yiying, ZHU Xiang, XU Naicai. Study on Mg-Al hydrotalcite modified by sodium dodecyl sulfate and its adsorption performance for Pb2+ [J]. Inorganic Chemicals Industry, 2025, 57(8): 48-57. |
| [10] | WANG Xiaoyu, DU Ruicheng, LI Yan, YANG Shuyan. Research advances in optimization and modification of TiO2-based nanomaterials [J]. Inorganic Chemicals Industry, 2025, 57(7): 24-34. |
| [11] | ZHAO Xueqing, WANG Zhonghui, WANG Yongwei, WANG Zhiyu, HUO Xiaodong. Study on preparation of nitrogen-doped porous carbon materials derived from used cigarette filters and their adsorption properties [J]. Inorganic Chemicals Industry, 2025, 57(7): 110-119. |
| [12] | YANG Hengyu, MO Hengliang, LI Tianyu, LÜ Long, CHEN Yili, WANG Luocong, ZHAO Wenfang, LIU Manman. Study on synthesis of high acid resistant Ti-based lithium ion sieve using mixed crystal TiO2 as titanium source [J]. Inorganic Chemicals Industry, 2025, 57(7): 57-63. |
| [13] | ZHANG Hanfei, SHEN Hongyan, LIU Youzhi. Study on preparation of MG composite material and its adsorption properties for methyl orange [J]. Inorganic Chemicals Industry, 2025, 57(6): 100-107. |
| [14] | PANG Hao, LI Bin, SUN Zhenhai, GUO Jian, CAI Zhe, WU Luming, HONG Meihua, YANG Zhanfeng. Study on adsorption and denitrification performance of iron doped modified SiO2 [J]. Inorganic Chemicals Industry, 2025, 57(6): 49-55. |
| [15] | CHEN Tongtong, HAN Xianying, JIE Mengling, LI Jiangang. Synergistically enhanced properties of Na4Fe3(PO4)2P2O7/C material by Mo-Mn dual doping and carbon nanotube composite [J]. Inorganic Chemicals Industry, 2025, 57(5): 79-86. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||
|
||