Inorganic Chemicals Industry ›› 2026, Vol. 58 ›› Issue (2): 68-75.doi: 10.19964/j.issn.1006-4990.2025-0018
• Environment·Health·Safety • Previous Articles Next Articles
Study on preparation of biochar via pyrolysis of acid-base synergistically treated chicken manure and its adsorption performance for Cd²⁺
HONG Menghuan1( ), NASEN Bate2, WANG Yaya2, LI Shuangqin1, XU Dehua1, YAN Zhengjuan1, LIU Wei2, WANG Xinlong1,2(
), NASEN Bate2, WANG Yaya2, LI Shuangqin1, XU Dehua1, YAN Zhengjuan1, LIU Wei2, WANG Xinlong1,2( )
)
- 1.Ministry of Education Research Center for Comprehensive Utilization and Clean Processing Engineering of Phosphorus Resources,School of Chemical Engineering,Sichuan University,Chengdu 610065,China
2.Xinjiang Key Laboratory of Clean Conversion and High-Value Utilization of Biomass Resources,School of Chemistry and Chemical Engineering,Yili Normal University,Yili 835000,China
-
Received:2025-01-06Online:2026-02-10Published:2026-03-12 -
Contact:WANG Xinlong E-mail:hmhhuan@163.com;wangxl@scu.edu.cn
CLC Number:
Cite this article
HONG Menghuan, NASEN Bate, WANG Yaya, LI Shuangqin, XU Dehua, YAN Zhengjuan, LIU Wei, WANG Xinlong. Study on preparation of biochar via pyrolysis of acid-base synergistically treated chicken manure and its adsorption performance for Cd²⁺[J]. Inorganic Chemicals Industry, 2026, 58(2): 68-75.
share this article
Table 2
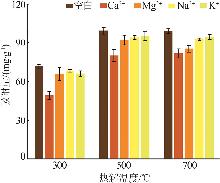
Main properties of biochar prepared by pyrolysis at different temperatures"
| 生物炭 | 产率/ % | 灰分/ % | w(C)/ % | w(H)/ % | w(N)/ % | w(O)/ % | H/C | O/C | w(K)/ % | w(Na)/ % | w(Ca)/ % | w(Mg)/ % | ID /IG |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| CMBC300 | 41.54 | 48.46 | 38.07 | 4.36 | 1.75 | 25.14 | 0.11 | 0.66 | 2.54 | 0.55 | 2.25 | 3.47 | 0.01 |
| CMBC500 | 31.24 | 56.23 | 31.95 | 2.11 | 0.92 | 20.07 | 0.07 | 0.63 | 2.65 | 0.59 | 2.39 | 3.61 | 2.15 |
| CMBC700 | 27.95 | 60.61 | 28.73 | 1.78 | 0.87 | 17.60 | 0.06 | 0.61 | 3.71 | 0.83 | 2.53 | 4.45 | 2.31 |
Table 4
Kinetic equqtion parameters of Cd2+adsorption by biochar"
| 生物炭 | 准一级动力学 | 准二级动力学 | Elovich | ||||||
|---|---|---|---|---|---|---|---|---|---|
k1/ min-1 | Qe/ (mg·g-1) | R2 | k2/(g·mg-1·min-1) | Qe/ (mg·g-1) | R2 | α/(mg·g-1·min-1) | β/ (g·mg-1) | R2 | |
| CMBC300 | 0.01 | 74.96 | 0.884 6 | 2.15×10-4 | 83.24 | 0.953 7 | 4.11 | 0.067 5 | 0.992 8 |
| CMBC500 | 0.03 | 88.27 | 0.782 0 | 4.66×10-4 | 93.84 | 0.901 1 | 24.95 | 0.076 9 | 0.975 6 |
| CMBC700 | 0.04 | 87.20 | 0.700 8 | 6.18×10-4 | 92.40 | 0.858 8 | 39.47 | 0.082 4 | 0.985 1 |
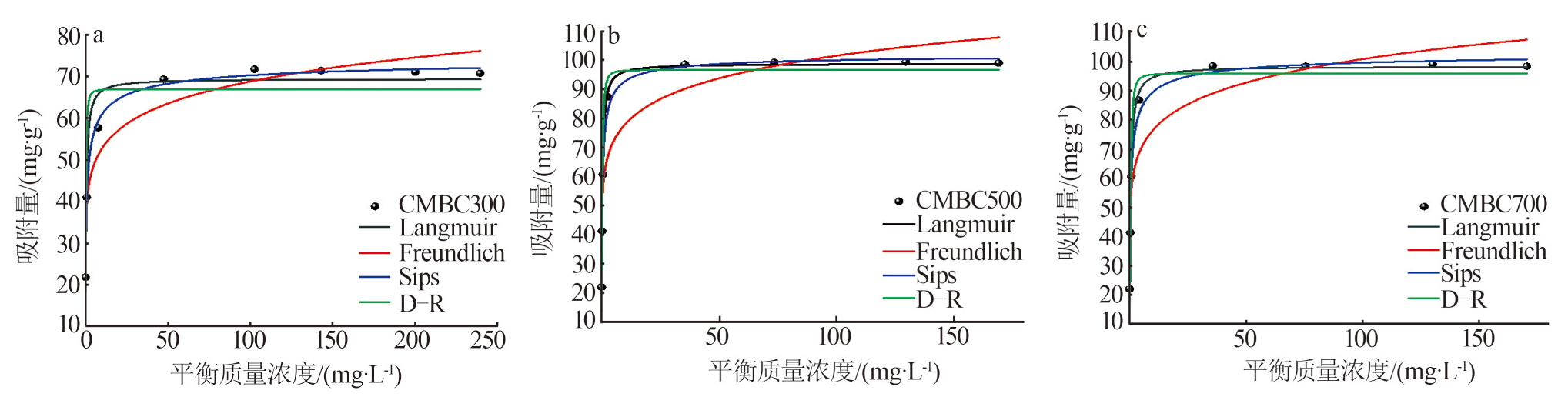
Table 5
Isotherm model parameters of Cd2+adsorption by biochar"
| 等温吸附模型 | CMBC300 | CMBC500 | CMBC700 | |
|---|---|---|---|---|
| Langmuir | Qm/(mg·g-1) | 69.38 | 98.66 | 98.27 |
| kL/(L·mg-1) | 2.20 | 3.55 | 2.49 | |
| R2 | 0.934 1 | 0.962 3 | 0.951 9 | |
| Freundlich | kF/(mg1-1/nF·L1/nF·g-1) | 40.37 | 59.80 | 57.91 |
| nF | 8.649 | 8.724 | 8.310 | |
| R2 | 0.908 3 | 0.853 9 | 0.871 8 | |
| Sips | Qm/(mg·g-1) | 75.38 | 102.31 | 103.9 |
| kS/(LnS·mgnS) | 1.287 | 2.260 | 1.692 | |
| nS | 0.512 0 | 0.627 9 | 0.565 4 | |
| R2 | 0.996 4 | 0.986 7 | 0.983 7 | |
| D-R | Qm/(mg·g-1) | 66.90 | 96.54 | 95.86 |
| β2/[mol2·(kJ-1)2] | 0.045 4 | 0.048 5 | 0.071 6 | |
| R2 | 0.824 9 | 0.915 9 | 0.900 5 | |
| [1] | 冯巍,程群鹏,李胜兰,等.鸡粪热解制备生物炭及其对废水中磷的吸附性能研究[J].武汉轻工大学学报,2021,40(1):75-80. |
| FENG Wei, CHENG Qunpeng, LI Shenglan,et al.Study on the preparation of biochar by pyrolysis of chicken manure and its adsorption performance for phosphorus in wastewater[J].Journal of Wuhan Polytechnic University,2021,40(1):75-80. | |
| [2] | 鞠天琛,於斯,李晓军,等.低温鸡粪生物质炭的制备及其对土壤理化性质的影响[J].吉林农业大学学报,2020,42(3):322-328. |
| JU Tianchen, YU Si, LI Xiaojun,et al.Low temperature preparation of biochar from chicken manure and its effect on soil physicochemical properties[J].Journal of Jilin Agricultural University,2020,42(3):322-328. | |
| [3] | RIGHI S, BANDINI V, MARAZZA D,et al.Life Cycle Assessment of high ligno-cellulosic biomass pyrolysis coupled with anaerobic digestion[J].Bioresource Technology,2016,212:245-253. |
| [4] | YUAN Xinsong, HE Tao, CAO Hongliang,et al.Cattle manure pyrolysis process:Kinetic and thermodynamic analysis with isoconversional methods[J].Renewable Energy,2017,107:489-496. |
| [5] | 王维猛,武卫兵,冯季军.阴离子交换膜辅助电解还原法处理含镉废水:原理及优化[J].环境化学,2022,41(12):3955-3964. |
| WANG Weimeng, WU Weibing, FENG Jijun.Anion exchange membrane-assisting electrochemical reduction treatment on Cd-containing wastewater:Principle and optimization[J].Environmental Chemistry,2022,41(12):3955-3964. | |
| [6] | KIBRIA K Q, ISLAM M A, HOQUE S,et al.Variations in cadmium accumulation among amon rice cultivars in Bangladesh and associated human health risks[J].Environmental Science and Pollution Research,2022,29(26):39888-39902. |
| [7] | TOFIGHY M A, MOHAMMADI T.Adsorption of divalent heavy metal ions from water using carbon nanotube sheets[J].Journal of Hazardous Materials,2011,185(1):140-147. |
| [8] | CHA J S, PARK S, JUNG S,et al.Production and utilization of biochar:A review[J].Journal of Industrial and Engineering Chemistry,2016,40:1-15. |
| [9] | CAI Tong, DU Huihui, LIU Xiaoli,et al.Insights into the removal of Cd and Pb from aqueous solutions by NaOH-EtOH-modified biochar[J].Environmental Technology & Innovation,2021,24:102031. |
| [10] | LI Bing, YANG Lan, WANG Changquan,et al.Adsorption of Cd(Ⅱ) from aqueous solutions by rape straw biochar derived from different modification processes[J].Chemosphere,2017,175:332- 340. |
| [11] | YANG Wen, SHANG Jianying, LI Baoguo,et al.Surface and colloid properties of biochar and implications for transport in porous media[J].Critical Reviews in Environmental Science and Technology,2020,50(23):2484-2522. |
| [12] | 郜礼阳,邓金环,唐国强,等.不同温度桉树叶生物炭对Cd2+的吸附特性及机制[J].中国环境科学,2018,38(3):1001-1009. |
| GAO Liyang, DENG Jinhuan, TANG Guoqiang,et al.Adsorption characteristics and Mechanism of Cd2+ on biochar with different pyrolysis temperatures produced from eucalyptus leaves[J].China Environmental Science,2018,38(3):1001-1009. | |
| [13] | 曹健华,刘凌沁,黄亚继,等.原料种类和热解温度对生物炭吸附Cd2+的影响[J].化工进展,2019,38(9):4183-4190. |
| CAO Jianhua, LIU Lingqin, HUANG Yaji,et al.Effects of feedstock type and pyrolysis temperature on Cd2+ adsorption by biochar[J].Chemical Industry and Engineering Progress,2019,38(9):4183-4190. | |
| [14] | 周蕾,陈兆兰,严玉波,等.鸡粪生物炭吸附固定铅的研究[J].中国农业科技导报,2022,24(11):199-207. |
| ZHOU Lei, CHEN Zhaolan, YAN Yubo,et al.Application of chicken manure-derived biochar for adsorption of lead[J].Journal of Agricultural Science and Technology,2022,24(11):199-207. | |
| [15] | LEE J I, CHOI D, KIM S,et al.Developing a sorptive material of cadmium from pyrolysis of hen manure[J].Chemosphere,2024,351:141262. |
| [16] | KIM W K, SHIM T, KIM Y S,et al.Characterization of cadmium removal from aqueous solution by biochar produced from a giant Miscanthus at different pyrolytic temperatures[J].Bioresource Technology,2013,138:266-270. |
| [17] | 康彩艳,李秋燕,刘金玉,等.不同热解温度生物炭对Cd2+的吸附影响[J].工业水处理,2021,41(5):68-72,79. |
| KANG Caiyan, LI Qiuyan, LIU Jinyu,et al.Effect of biochar at different pyrolysis temperatures on the adsorption of Cd2+ [J].Industrial Water Treatment,2021,41(5):68-72,79. | |
| [18] | WANG Shengsen, GAO Bin, ZIMMERMAN A R,et al.Removal of arsenic by magnetic biochar prepared from pinewood and natural hematite[J].Bioresource Technology,2015,175:391-395. |
| [19] | MASZKOWSKA J, WAGIL M, MIODUSZEWSKA K,et al.Thermodynamic studies for adsorption of ionizable pharmaceuticals onto soil[J].Chemosphere,2014,111:568-574. |
| [20] | 宋朝霞,刘永康,郭耀坤,等.小麦秸秆生物炭对孔雀石绿吸附性能的研究[J].无机盐工业,2024,56(9):128-135. |
| SONG Zhaoxia, LIU Yongkang, GUO Yaokun,et al.Study on adso-rption performance of wheat straw biochar on malachite green[J].Inorganic Chemicals Industry,2024,56(9):128-135. |
| [1] | WU Jie, XU Chunhui, WANG Feng, TANG Zhongfeng. Effect of NaNO3 doping on structure and adsorption-desorption performance of MgO-based CO2 adsorbents [J]. Inorganic Chemicals Industry, 2026, 58(2): 60-67. |
| [2] | WU Fenglong, SONG Jin. Study on photocatalytic synthesis of low molecular weight anti⁃mud polycarboxylate superplasticizers and its adsorption effect for kaolin [J]. Inorganic Chemicals Industry, 2026, 58(1): 52-60. |
| [3] | WANG Guibin, ZHANG Hongwei, LIU Lijuan, ZHAO Yu, ZHANG Dongqiang. Study on effect of resin modification on removal performance of fluoride and chloride ions in desulfurization ionic liquid [J]. Inorganic Chemicals Industry, 2026, 58(1): 18-25. |
| [4] | CHEN Jianjun, LI Li, LIU Laibao, ZHANG Daiyu, JIANG Chenxi, ZHU Hanzhen, WANG Fu, LIAO Qilong. Study on synthesis of high specific surface area calcium hydroxide by quicklime digestion and its adsorption performance for lead [J]. Inorganic Chemicals Industry, 2026, 58(1): 99-107. |
| [5] | WANG Zhigang, HU Xiaodong, WANG Hui, XUE Lianggang, TIAN Jianqiang. Study on preparation of NO2- modified hydrotalcite and its corrosion resistance to steel bars [J]. Inorganic Chemicals Industry, 2025, 57(8): 74-81. |
| [6] | HAN Xing, SHI Dandan, WANG Xingquan, ZHANG Tianying, CAO Yue, WANG Yiying, ZHU Xiang, XU Naicai. Study on Mg-Al hydrotalcite modified by sodium dodecyl sulfate and its adsorption performance for Pb2+ [J]. Inorganic Chemicals Industry, 2025, 57(8): 48-57. |
| [7] | ZHAO Xueqing, WANG Zhonghui, WANG Yongwei, WANG Zhiyu, HUO Xiaodong. Study on preparation of nitrogen-doped porous carbon materials derived from used cigarette filters and their adsorption properties [J]. Inorganic Chemicals Industry, 2025, 57(7): 110-119. |
| [8] | YANG Jingjing, XU Chenchen, ZHU Liyan, KUAI Qiang, WU Bingdang, HUANG Tianyin. Study on adsorption and removal of nitroimidazole antibiotics in water by potassium carbonate-activated algae-based biochar [J]. Inorganic Chemicals Industry, 2025, 57(7): 99-109. |
| [9] | YANG Hengyu, MO Hengliang, LI Tianyu, LÜ Long, CHEN Yili, WANG Luocong, ZHAO Wenfang, LIU Manman. Study on synthesis of high acid resistant Ti-based lithium ion sieve using mixed crystal TiO2 as titanium source [J]. Inorganic Chemicals Industry, 2025, 57(7): 57-63. |
| [10] | ZHANG Hanfei, SHEN Hongyan, LIU Youzhi. Study on preparation of MG composite material and its adsorption properties for methyl orange [J]. Inorganic Chemicals Industry, 2025, 57(6): 100-107. |
| [11] | PANG Hao, LI Bin, SUN Zhenhai, GUO Jian, CAI Zhe, WU Luming, HONG Meihua, YANG Zhanfeng. Study on adsorption and denitrification performance of iron doped modified SiO2 [J]. Inorganic Chemicals Industry, 2025, 57(6): 49-55. |
| [12] | SUN Mengyuan, HE Wei, WANG Chen. Research progress of preparation and adsorption of magnesium oxide fibers and their composite fibers [J]. Inorganic Chemicals Industry, 2025, 57(5): 1-10. |
| [13] | BAI Xingxing, LI Hanfei, TANG Yong, ZHANG Jun, ZHU Guangkai, LI Lishuo, TONG Zhangfa. Study on preparation of cellulose based hydrogel doped with nano-calcium carbonate and its adsorption properties of copper ions [J]. Inorganic Chemicals Industry, 2025, 57(2): 83-91. |
| [14] | HOU Daiwen, LIANG Huan, XING Lijing, LIU Hai, ZHANG Huali, TIAN Chengtao, SHI Tongshan. Study on preparation and phosphorus adsorption of phosphate tailings ceramic particles by response surface method [J]. Inorganic Chemicals Industry, 2025, 57(11): 74-82. |
| [15] | JIN Zhouzheng, ZHANG Donghui, PENG Xueping, LIN Minyan, DAI Zhongyuan, CHEN Changhua. Research progress on typical calcium-based industrial solid waste mineralization process and utilization in building materials [J]. Inorganic Chemicals Industry, 2025, 57(10): 11-23. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||
|
||