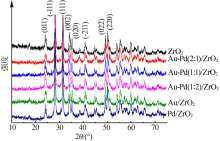
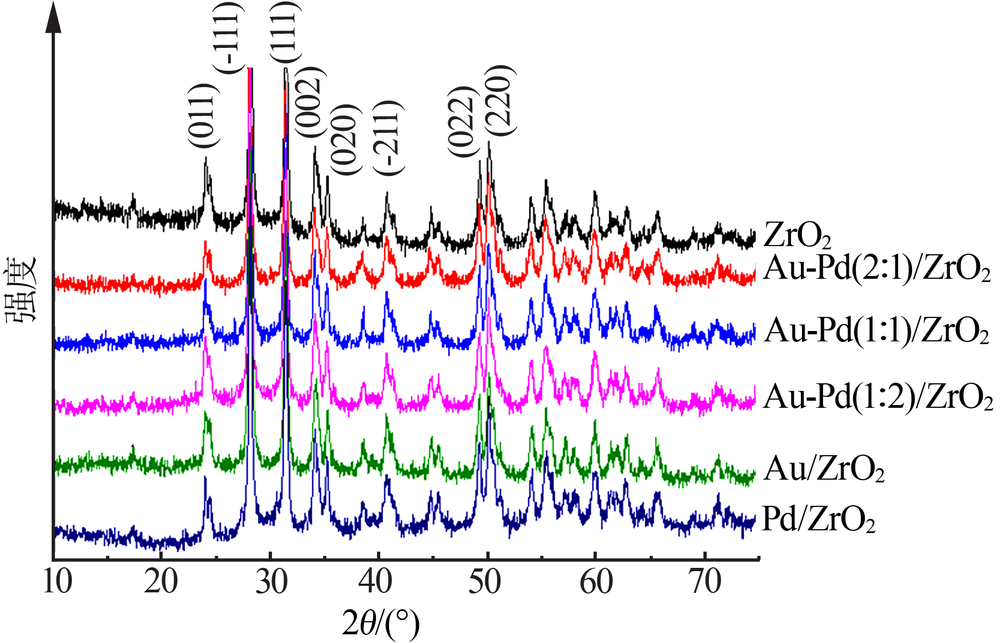
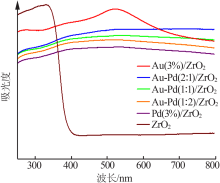
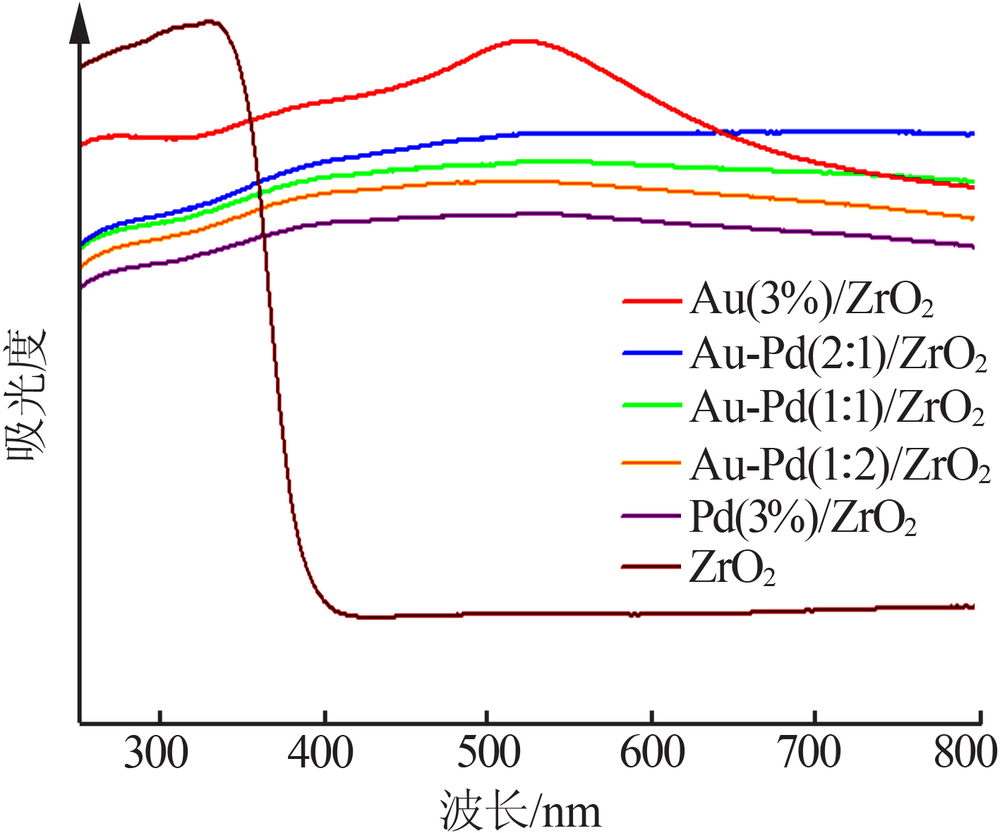
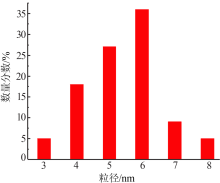
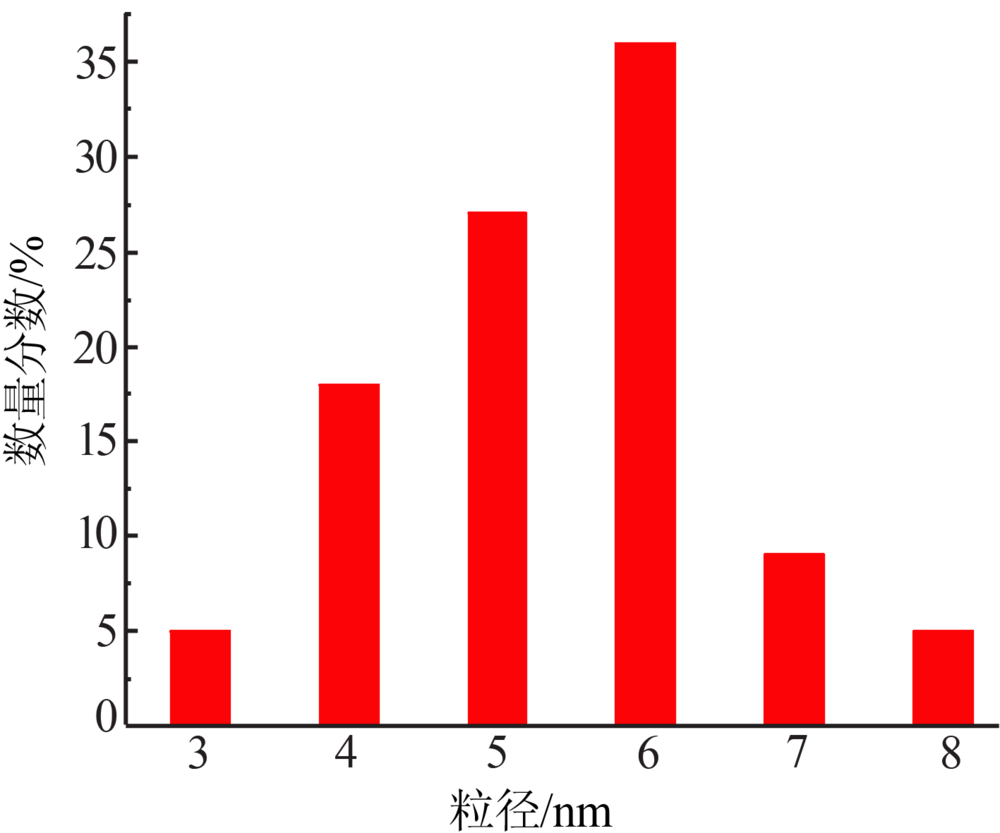
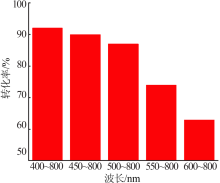
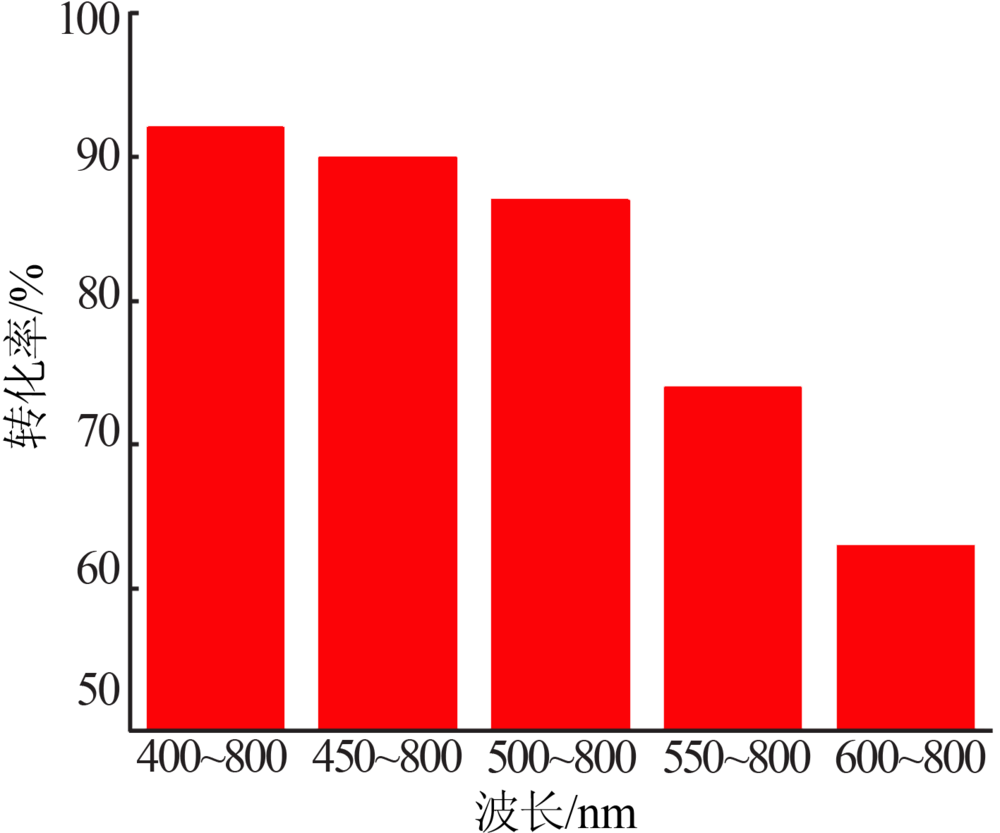
| [1] |
Huang G, Luo J, Deng C C , et al. Catalytic oxidation of toluene with molecular oxygen over mangmanese tetraphenlyporphyrin supported on chitosan[J]. Applied Catalysis A:General, 2008,338(1/2):83-86.
doi: 10.1016/j.apcata.2007.12.027
|
| [2] |
Mitsudome T, Mikami Y, Funai H , et al. Oxidant-free alcohol dehy-drogenation using a reusable hydrotalcite-supported silver nano-particles catalyst[J]. Angew.Chem.Int.Ed., 2007,47(1):138-141.
|
| [3] |
Zhao G, Deng M, Jiang Y , et al. Microstructured Au/Ni-fiber cata-lyst;galvanic reaction preparation and catalytic performance for low-temperature gas-phase alcohol oxidation[J]. Journal of Cataly-sis, 2013,301:46-53.
|
| [4] |
Fan J, Dai Y, Li Y , et al. Low-temperature,highly selective,gas-phase oxidation of benzyl alcohol over mesoporous K-Cu-TiO2 with stable copper(Ⅰ) oxidation state[J]. Journal of the American Che-mical Society, 2009,131(43):15568-15569.
doi: 10.1021/ja9032499
pmid: 19860474
|
| [5] |
Bond G C, Sermon P A . Gold catalysts for olefin hydrogenation[J]. Gold Bull., 1973,6(4):102-105.
|
| [6] |
Haruta M, Kobayashi T, Sano H , et al. Novel gold catalysts for the oxidation of carbon monoxide at a temperature far below 0 ℃[J]. Chem Lett., 1987,16(2):405-408.
|
| [7] |
Hutchings G J . Vapor phase hydorchlorination of acetylene:correla-tion of catalytic activity of supported metal chloride catalysts[J]. J.Catal., 1985,96(1):292-295.
|
| [8] |
Kawabata T, Shinozuka Y, Ohishi Y , et al. Nickel containing Mg-Al hydrotalcite-type anionic clay catalyst for the oxidation of alcohols with molecular oxygen[J]. J.Mol.Catal.A:Chem., 2005,236(1):206-215.
|
| [9] |
Pina C D, Falletta E, Rossi M . Update on selective oxidation using gold[J]. Chem.Soc.Rev., 2012,41(1):350-369.
doi: 10.1039/c1cs15089h
pmid: 21727977
|
| [10] |
McEwan L, Julius M, Roberts S , et al. A review of the use of gold ca-talysts in selective hydrogenation reactions Lynsey McEwana[J]. Gold Bull., 2010,43(4):298-306.
doi: 10.1007/BF03214999
|
| [11] |
Yang X F, Wang A Q, Wang Y L , et al. Unusual selectivity of gold catalysts for hydrogenation of1,3-butadiene toward cis-2-butene:A joint experimental and theoretical investigation[J]. J.Phys.Chem.C, 2010,114(7):3131-3139.
|
| [12] |
Shimizu K, Sato R, Satsuma A . Direct C-C cross-coupling of secon-dary and primary alcohols catalyzed by a gamma-alumina-suppo-rted silver subnanocluster[J]. Angew.Chem.Int.Ed., 2009,48:3982-3986.
|
| [13] |
Chen X, Zheng Z, Ke X , et al. Supported silver nanoparticles as photocatalysts under ultraviolet and visible light irradiation[J]. Green Chem., 2010,12(3):414-419.
doi: 10.1039/b921696k
|
| [14] |
Fang W H, Chen J S, Zhang Q H , et al. Hydrotalcite-supported gold catalyst for the oxidant-free dehydrogenation of benzyl alcohol:studies on support and gold size effects[J]. Chemistry A European Journal, 2011,17(4):1247-1256.
doi: 10.1002/chem.201002469
pmid: 21243691
|
| [15] |
Shao M W, Wang H, Zhang M L , et al. The mutual promotional ef-fect of Au-Pd bimetallic nanoparticles on silicon nanowires:A study of preparation and catalytic activity[J]. Appl.Phys.Lett., 2008,93:243110.
|
| [16] |
Schunemann S, van Gastel M, Tüysüz H . A CsPbBr3/TiO2 compo-site for visible-light-driven photocatalytic benzyl alcohol oxidation[J]. ChemSusChem, 2018,11:2057-2061.
doi: 10.1002/cssc.201800679
pmid: 29790659
|
| [17] |
Song T, Park J E, Chung Y K . Rhodium-catalyzed synjournal of im-ines and esters from benzyl alcohols and nitroarenes:Chang in ca-talyst reactivity depending on the presence or absence of the pho-shine ligand[J]. The Journal of Organic Chemistry, 2018,83(7):4197-4203.
doi: 10.1021/acs.joc.8b00197
pmid: 29536727
|
| [18] |
Wu J B, Shi R P, Qin Z F , et al. Selective oxidation of methanol to methyl formate over bimetallic Au-Pd nanoparticles supported on SiO2[J]. Journal of Fuel Chemistry and Technology, 2019,47(7):780-790.
|
 )
)