Inorganic Chemicals Industry ›› 2021, Vol. 53 ›› Issue (7): 23-29.doi: 10.19964/j.issn.1006-4990.2021-0008
• Reviews and Special Topics • Previous Articles Next Articles
Research progress rational design of noble metal catalysts for selective hydrogenation
Ma Mingchao1,2( ),Zang Jiazhong1,2(
),Zang Jiazhong1,2( ),Yu Haibin1,2,Fan Jingxin1,2,Guo Chunlei1,2,Jin Fengying1,2
),Yu Haibin1,2,Fan Jingxin1,2,Guo Chunlei1,2,Jin Fengying1,2
- 1. CenerTech Tianjin Chemical Research and Design Institute Ltd.,Co.,Tianjin 300131,China
2. Tianjin Refining and Catalytic Technology Engineering Center
-
Received:2021-01-05Online:2021-07-10Published:2021-07-13 -
Contact:Zang Jiazhong E-mail:mamingchao0829@163.com;zangjiazhong@163.com
CLC Number:
Cite this article
Ma Mingchao,Zang Jiazhong,Yu Haibin,Fan Jingxin,Guo Chunlei,Jin Fengying. Research progress rational design of noble metal catalysts for selective hydrogenation[J]. Inorganic Chemicals Industry, 2021, 53(7): 23-29.
share this article
| [1] | Liu L C, Corma A. Metal catalysts for heterogeneous catalysis:From single atoms to nanoclusters and nanoparticles[J]. Chemical Revie-ws, 2018, 118(10):4981-5079. |
| [2] |
Somorjai G A, Yang M. The surface science of catalytic selectivi-ty[J]. Topics in Catalysis, 2003, 24(1):61-72.
doi: 10.1023/B:TOCA.0000003077.98309.cf |
| [3] | Gertl H K, Knözinger H. Handbook of heterogeneous catalysis[M]. New York:Wiley-VCH, 1997:46-48. |
| [4] | 朱利安R.H.罗斯. 多相催化:基本原理与应用[M]. 田野,张立红,赵宜成,等译. 北京: 化学工业出版社, 2016. |
| [5] |
Somorjai G A, Park J Y. Molecular factors of catalytic selectivity[J]. Angewandte Chemie International Edition, 2008, 47:9212-9228.
doi: 10.1002/anie.200803181 |
| [6] |
Zhang S, Chang C R, Huang Z Q, et al. High catalytic activity and chemoselectivity of sub-nanometric Pd clusters on porous nanorods of CeO2 for hydrogenation of nitroarenes[J]. Journal of the American Chemical Society, 2016, 138(8):2629-2637.
doi: 10.1021/jacs.5b11413 |
| [7] |
Wu B, Zheng N F. Surface and interface control of noble metal nanocrystals for catalytic and electrocatalytic applications[J]. Nano Today, 2013, 8:168-197.
doi: 10.1016/j.nantod.2013.02.006 |
| [8] | Ye T N, Xiao Z, Li J, et al. Stable single platinum atoms trapped in sub-nanometer cavities in 12CaO·7Al2O3 for chemoselective hydro-genation of nitroarenes[J]. Nature Communications, 2020, 11.Doi: 10.1038/s41467-019-14216-9. |
| [9] |
Serna P, Boronat M, Corma A. Tuning the behavior of Au and Pt cat- alysts for the chemoselective hydrogenation of nitroaromatic compo-unds[J]. Topics in Catalysis, 2011, 54(5/6/7):439-446.
doi: 10.1007/s11244-011-9668-z |
| [10] | Corma A, Serna P, Concepción P, et al. Transforming nonselective into chemoselective metal catalysts for the hydrogenation of sub- stituted nitroaromatics[J]. Journal of the American Chemical So-ciety, 2008, 130(27):8748-8753. |
| [11] |
Huang W, Sun G, Cao T. Surface chemistry of group IB metals and related oxides[J]. Chemical Society Reviews, 2017, 46:1977-2000.
doi: 10.1039/C6CS00828C |
| [12] |
Zhao M, Yuan K, Wang Y, et al. Metal-organic frameworks as se-lectivity regulators for hydrogenation reactions[J]. Nature, 2016, 539:76-80.
doi: 10.1038/nature19763 |
| [13] | Lu J, Low K B, Lei Y, et al. Toward atomically-precise synjournal of supported bimetallic nanoparticles using atomic layer deposi-tion[J]. Nature Communications, 2014, 5.Doi: 10.1038/ncomms4264. |
| [14] |
Nϕrskov J K, Bligaard T, Rossmeisl J, et al. Towards the computational design of solid catalysts[J]. Nature Chemistry, 2009, 1(1):37-46.
doi: 10.1038/nchem.121 |
| [15] |
Noyori R. Synthesizing our future[J]. Nature Chemistry, 2009, 1(1):5-6.
doi: 10.1038/nchem.143 pmid: 21378778 |
| [16] | Ertl G. Heterogeneous catalysis on the atomic scale[J]. The Chemi-cal Record, 2001, 1(1):33-45. |
| [17] | Boudart M. Heterogeneous catalysis by metals[J]. Journal of Molec-ular Catalysis, 1985, 30:27-38. |
| [18] | Boudart M. Catalysis by supported metals[J]. Advances in Cataly-sis, 1969, 20:153-166. |
| [19] |
Boronat M, Leyva-Pérez A, Corma A. Theoretical and experimental insights into the origin of the catalytic activity of subnanometric gold clusters:Attempts to predict reactivity with clusters and nano-particles of gold[J]. Accounts of Chemical Research, 2014, 47(3):834-844.
doi: 10.1021/ar400068w |
| [20] |
Gerhard E. Heterogeneous catalysis on atomic scale[J]. Journal of Molecular Catalysis A:Chemical, 2002, 182/183:5-16.
doi: 10.1016/S1381-1169(01)00460-5 |
| [21] |
Chen G, Xu C, Huang X, et al. Interfacial electronic effects control the reaction selectivity of platinum catalysts[J]. Nature Materials, 2016, 15:564-569.
doi: 10.1038/nmat4555 |
| [22] | Novotny Z, Argentero G, Wang Z M, et al. Ordered array of single Au adatoms with remarkable thermal stability:Au/Fe3O4(001)[J]. Physical Review Letters, 2012, 108.Doi: 10.1103/PhysRevLett.108.216103. |
| [23] |
Ferguson G A, Yin C R, Kwon G, et al. Stable subnanometer cobalt oxide clusters on ultrananocrystalline diamond and alumina supports:Oxidation state and the origin of sintering resistance[J]. The Journal of Physical Chemistry C, 2012, 116(45):24027-24034.
doi: 10.1021/jp3041956 |
| [24] |
Bell A T. The impact of nanoscience on heterogeneous catalysis[J]. Science, 2003, 299:1688-1691.
doi: 10.1126/science.1083671 |
| [25] |
Chen M S, Goodman D W. The structure of catalytically active gold on titania[J]. Science, 2004, 306:252-255.
doi: 10.1126/science.1102420 |
| [26] |
Hansen T W, DeLaRiva A T, Challa S R, et al. Sintering of catalytic nanoparticles:Particle migration or Ostwald ripening?[J]. Accounts of Chemical Research, 2013, 46(8):1720-1730.
doi: 10.1021/ar3002427 |
| [27] |
Sanchez S I, Menard L D, Bram A, et al. The emergence of nonbulk properties in supported metal clusters:Negative thermal expansion and atomic disorder in Pt nanoclusters supported on γ-Al2O3[J]. Journal of the American Chemical Society, 2009, 131(20):7040-7054.
doi: 10.1021/ja809182v |
| [28] | Yang X F, Wang A Q, Qiao B T, et al. Single-atom catalysts:A new frontier in heterogeneous catalysis[J]. Accounts of Chemical Re-search, 2013, 46(8):1740-1748. |
| [29] | Bao X H. Fundamental research in catalysis with emphasis on con-finement effects[J]. Scientia Sinica Chimica, 2012, 42(4).Doi: 10.1360/032012-130. |
| [30] | Li H, Xiao J, Fu Q, et al. Confined catalysis under two-dimensional materials[J]. Proceedings of the National Academy of Sciences of the United States of America, 2017, 114:5930-5934. |
| [31] |
Dejong K P, Zecevic J. The platinum tush[J]. Nature Materials, 2017, 16:7-8.
doi: 10.1038/nmat4832 |
| [32] |
Corma A, Serna P. Chemoselective hydrogenation of nitro compoun-ds with supported gold catalysts[J]. Science, 2006, 313:332-334.
doi: 10.1126/science.1128383 |
| [33] |
Bhogeswararao S, Srinivas D. Intramolecular selective hydrogena-tion of cinnamaldehyde over CeO2-ZrO2 supported Pt catalysts[J]. Journal of Catalysis, 2012, 285:31-40.
doi: 10.1016/j.jcat.2011.09.006 |
| [34] |
Shi W, Zhang B S, Lin Y M, et al. Enhanced chemoselective hydro-genation through tuning the interaction between Pt nanoparticles and carbon supports:Insights from identical location transmission electron microscopy and X-ray photoelectron spectroscopy[J]. ACS Catalysis, 2016, 6(11):7844-7854.
doi: 10.1021/acscatal.6b02207 |
| [35] |
Pan H, Li J, Lu J, et al. Selective hydrogenation of cinnamaldehyde with PtFeOx/Al2O3@SBA-15 catalyst:Enhancement in activity and selectivity to unsaturated alcohol by Pt-FeOx and Pt-Al2O3@SBA-15 interaction[J]. Journal of Catalysis, 2017, 354:24-36.
doi: 10.1016/j.jcat.2017.07.026 |
| [36] |
Shu Y, Chan H C, Xie L, et al. Bimetallic platinum-tin nanoparti-cles on hydrogenated molybdenum oxide for the selective hydro-genation of functionalized nitroarenes[J]. ChemCatChem, 2017, 9:4199-4205.
doi: 10.1002/cctc.201700880 |
| [37] | Wang A, Li J, Zhang T. Heterogeneous single-atom catalysis[J]. Nature Reviews Chemistry, 2018(2):65-81. |
| [38] |
Wang C P, Mao S J, Wang Z, et al. Insight into single-atom-induced unconventional size dependence over CeO2-supported Pt catalys-ts[J]. Chem, 2020, 6(3):752-765.
doi: 10.1016/j.chempr.2019.12.029 |
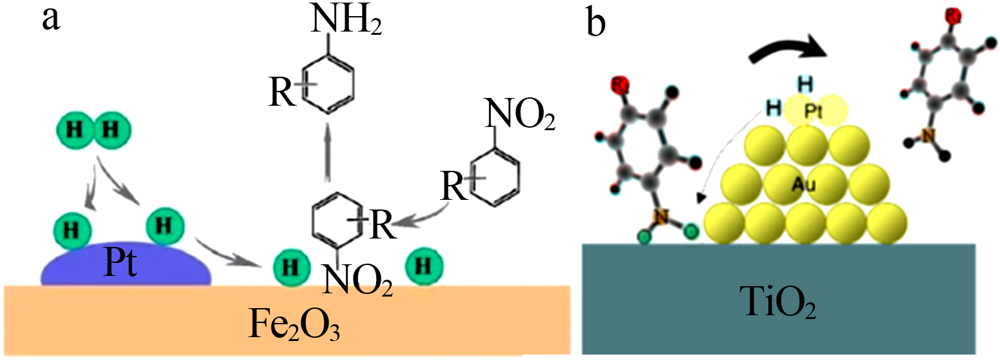
| [39] | Wei H, Liu X, Wang A, et al. FeOx-supported platinum single-atom and pseudo-single-atom catalysts for chemoselective hydrogena-tion of functionalized nitroarenes[J]. Nature Communications, 2014, 5.Doi: 10.1038/ncomms6634. |
| [40] |
Feng H, Lu J L, Stair P C, et al. Alumina over-coating on Pd nano-particle catalysts by atomic layer deposition:Enhanced stability and reactivity[J]. Catalysis Letters, 2011, 141(4):512-517.
doi: 10.1007/s10562-011-0548-8 |
| [41] | Hu Q, Wang S, Gao Z, et al. The precise decoration of Pt nanopar-ticles with Fe oxide by atomic layer deposition for the selective hydrogenation of cinnamaldehyde[J]. Applied Catalysis B:Enviro-nmental, 2017, 218:591-599. |
| [42] |
He T W, Zhang C M, Zhang L, et al. Single Pt atom decorated graphitic carbon nitride as an efficient photocatalyst for the hydrogenation of nitrobenzene into aniline[J]. Nano Research, 2019, 12(8):1817-1823.
doi: 10.1007/s12274-019-2439-z |
| [43] |
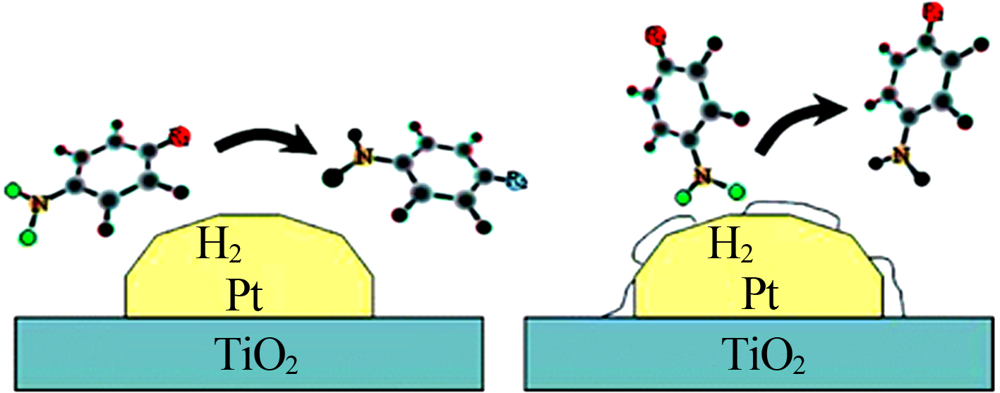
Cárdenas Lizana F, Hao Y F, Crespo Quesada M, et al. Selective gas phase hydrogenation of p-chloronitrobenzene over Pd catalysts:Role of the support[J]. ACS Catalysis, 2013, 3(6):1386-1396.
doi: 10.1021/cs4001943 |
| [44] |
Mao S J, Zhao B W, Wang Z, et al. Tuning the catalytic performance for the semi-hydrogenation of alkynols by selectively poisoning the active sites of Pd catalysts[J]. Green Chemistry, 2019, 21:4143-4151.
doi: 10.1039/C9GC01356C |
| [45] | 曾昭槐. 择形催化[M]. 北京: 中国石化出版社, 1994:145-153. |
| [46] |
Zhang J, Wang L, Shao Y, et al. A Pd@zeolite catalyst for nitroarene arene hydrogenation with high product selectivity by sterically controlled adsorption in the zeolite micropores[J]. Angewandte Chemie International Edition, 2017, 56:9747-9751.
doi: 10.1002/anie.v56.33 |
| [47] |
Guo Z Y, Xiao C X, Maligal Ganesh R V, et al. Pt nanoclusters confined within metal-organic framework cavities for chemoselec-tive cinnamaldehyde hydrogenation[J]. ACS Catalysis, 2014, 4(5):1340-1348.
doi: 10.1021/cs400982n |
| [48] |
Li X F, Wang Z, Mao S J, et al. Insight into the role of additives in catalytic synjournal of cyclohexylamine from nitrobenzene[J]. Chinese Journal of Catalysis, 2018, 36:1191-1196.
doi: 10.1016/S1872-2067(15)60921-4 |
| [49] |
Chen Y Z, Kong X Q, Mao S J, et al. Study on the role of alkaline sodium additive in selective hydrogenation of phenol[J]. Chinese Journal of Catalysis, 2019, 40:1516-1524.
doi: 10.1016/S1872-2067(19)63386-3 |
| [50] |
Mao S J, Wang C P, Wang Y, et al. The chemical nature of N doping on N doped carbon supported noble metal catalysts[J]. Journal of Catalysis, 2019, 375:456-465.
doi: 10.1016/j.jcat.2019.06.039 |
| [51] |
Chen Y Z, Wang Z, Mao S J, et al. Rational design of hydrogenation catalysts using nitrogen-doped porous carbon[J]. Chinese Journal of Catalysis, 2019, 40:971-979.
doi: 10.1016/S1872-2067(19)63353-X |
| [52] |
Liu J R, Xie L, Wang Z, et al. Biomass-derived ordered mesoporous carbon nano-ellipsoid encapsulated metal nanoparticles inside:Ideal nanoreactors for shape-selective catalysis[J]. Chemical Communications, 2020, 56(2):229-232.
doi: 10.1039/C9CC08066J |
| [53] |
Jing P, Gan T, Qi H, et al. Synergism of Pt nanoparticles and iron oxide support for chemoselective hydrogenation of nitroarenes un-der mild conditions[J]. Chinese Journal of Catalysis, 2019, 40:214-222.
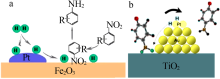
doi: 10.1016/S1872-2067(19)63276-6 |
| [54] |
Serna P, Concepción P, Corma A. Design of highly active and che-moselective bimetallic gold-platinum hydrogenation catalysts thr-ough kinetic and isotopic studies[J]. Journal of Catalysis, 2009, 265:19-25.
doi: 10.1016/j.jcat.2009.04.004 |
| [55] |
Chandler B D. An extra layer of complexity[J]. Nature Chemistry, 2017, 9:108-109.
doi: 10.1038/nchem.2724 pmid: 28282056 |
| [56] |
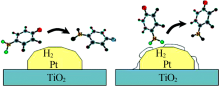
Dandekar A, Vannice M A. Crotonaldehyde hydrogenation on Pt/TiO2 and Ni/TiO2 SMSI catalysts[J]. Journal of Catalysis, 1999, 183:344-354.
doi: 10.1006/jcat.1999.2419 |
| [57] |
Concepción P, Corma A, Silvestre Albero J, et al. Chemoselective hydrogenation catalysts:Pt on mesostructured CeO2 nanoparticles embedded within ultrathin layers of SiO2 binder[J]. Journal of the American Chemical Society, 2004, 126(17):5523-5532.
doi: 10.1021/ja031768x |
| [58] |
Kennedy G, Baker L R, Somorjai G A. Selective amplification of CO bond hydrogenation on Pt/TiO2:Catalytic reaction and sum-frequ-ency generation vibrational spectroscopy studies of crotonaldehyde hydrogenation[J]. Angewandte Chemie International Edition, 2014, 53:3405-3408.
doi: 10.1002/anie.201400081 |
| [59] |
Kennedy G, Melaet G, Han H L, et al. In situ spectroscopic investi-gation into the active sites for crotonaldehyde hydrogenation at the Pt nanoparticle-Co3O4 interface[J]. ACS Catalysis, 2016, 6(10):7140-7147.
doi: 10.1021/acscatal.6b01640 |
| [1] | SONG Xiuduo,CAI Zhe,LI Xiaoyun,SUN Yanyin,HAN Enshan. Study on catalytic performance of Mg-MOF-74 in the Knoevenagel condensation reaction [J]. Inorganic Chemicals Industry, 2022, 54(2): 111-116. |
| [2] | JIN Fengying,YU Haibin,WANG Yaquan,ZANG Jiazhong,GUO Chunlei,LIU Hang,MA Mingchao,ZHAO Xunzhi,LIU Kailong. Effect of Ni on selective hydrodegenation of polyaromatic hydrocarbons of Pt/γ-Al2O3 catalyst [J]. Inorganic Chemicals Industry, 2022, 54(10): 141-148. |
| [3] | Zhang Shangqiang,Sun Guanhua,Sun Yanmin,Zhu Jinjian,Nan Jun,Xiao Han,Zhang Jingcheng,Song Guoliang. Study on preparation of CNTs-Al2O3 supported Pd catalyst and its performance [J]. Inorganic Chemicals Industry, 2021, 53(6): 194-198. |
| [4] | Liu Yuxin1,Shi Fengjuan2,Lü Yaohui1,Wei Shicheng1,Wang Yujiang1,He Dongyu1,Wang Wenyu1. Progress in preparation of nano?鄄sized or porous chromium oxide particles [J]. Inorganic Chemicals Industry, 2019, 51(10): 1-6. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||
|
||