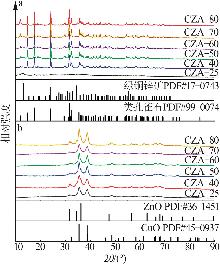
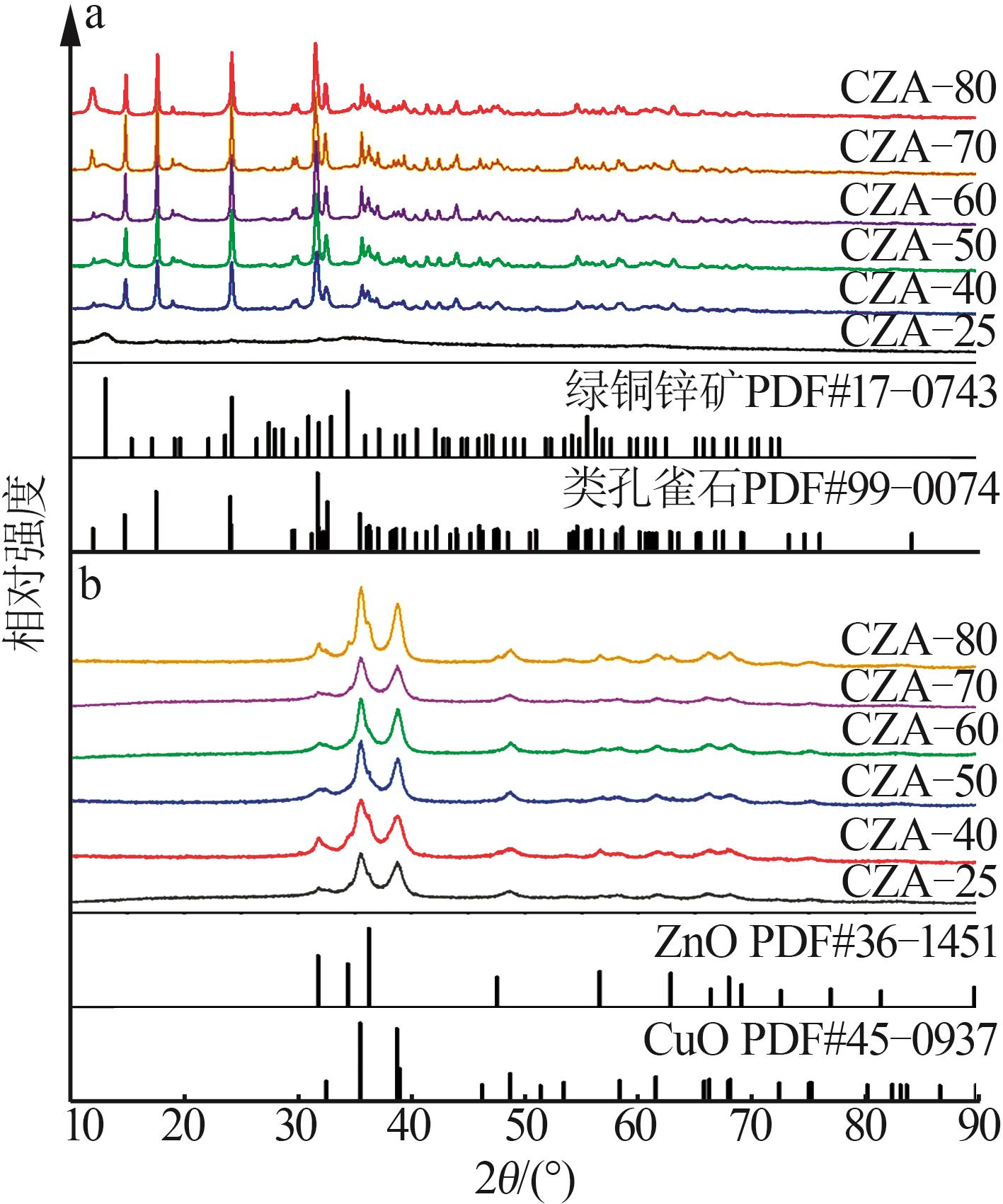
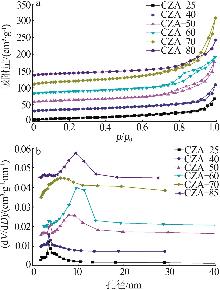
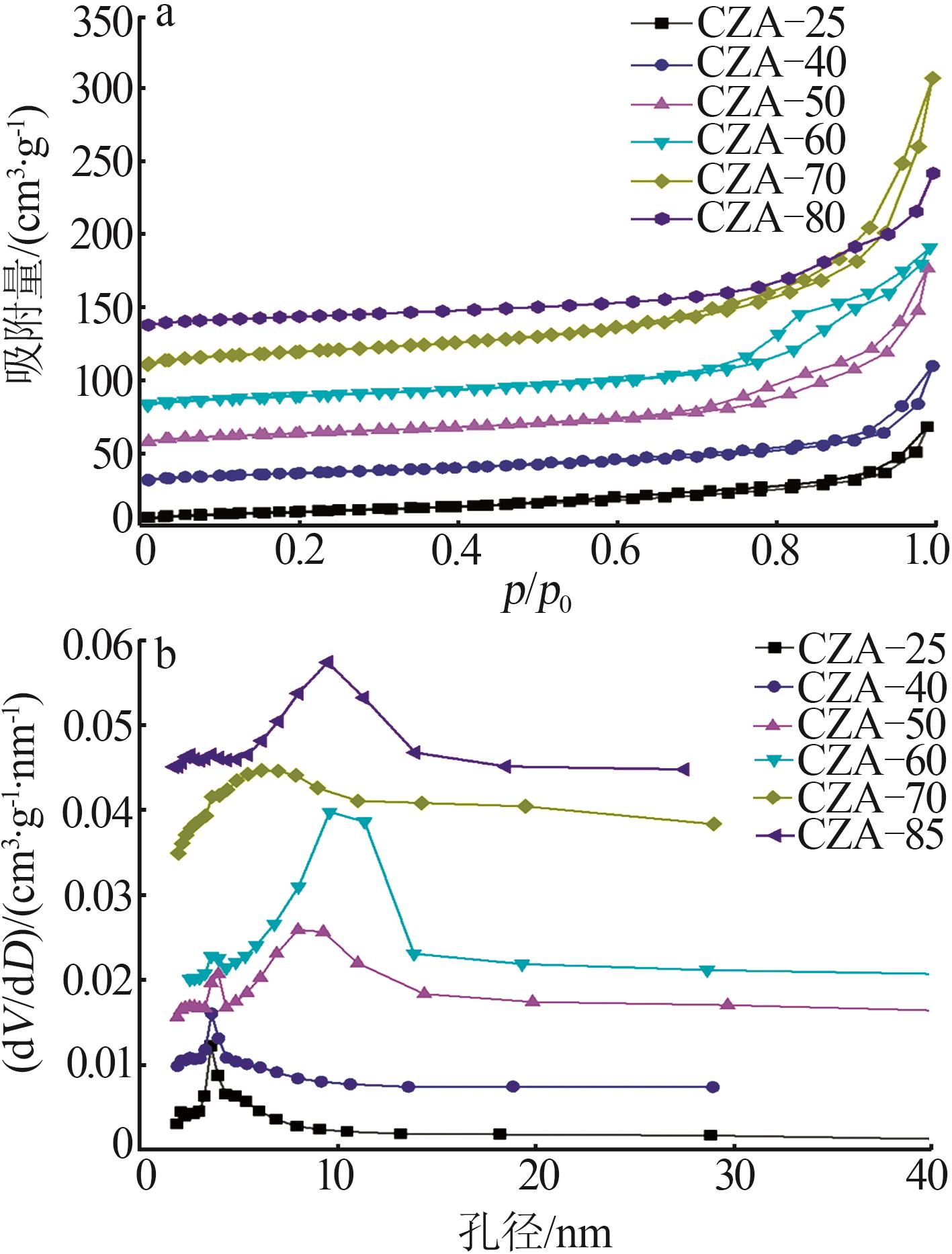
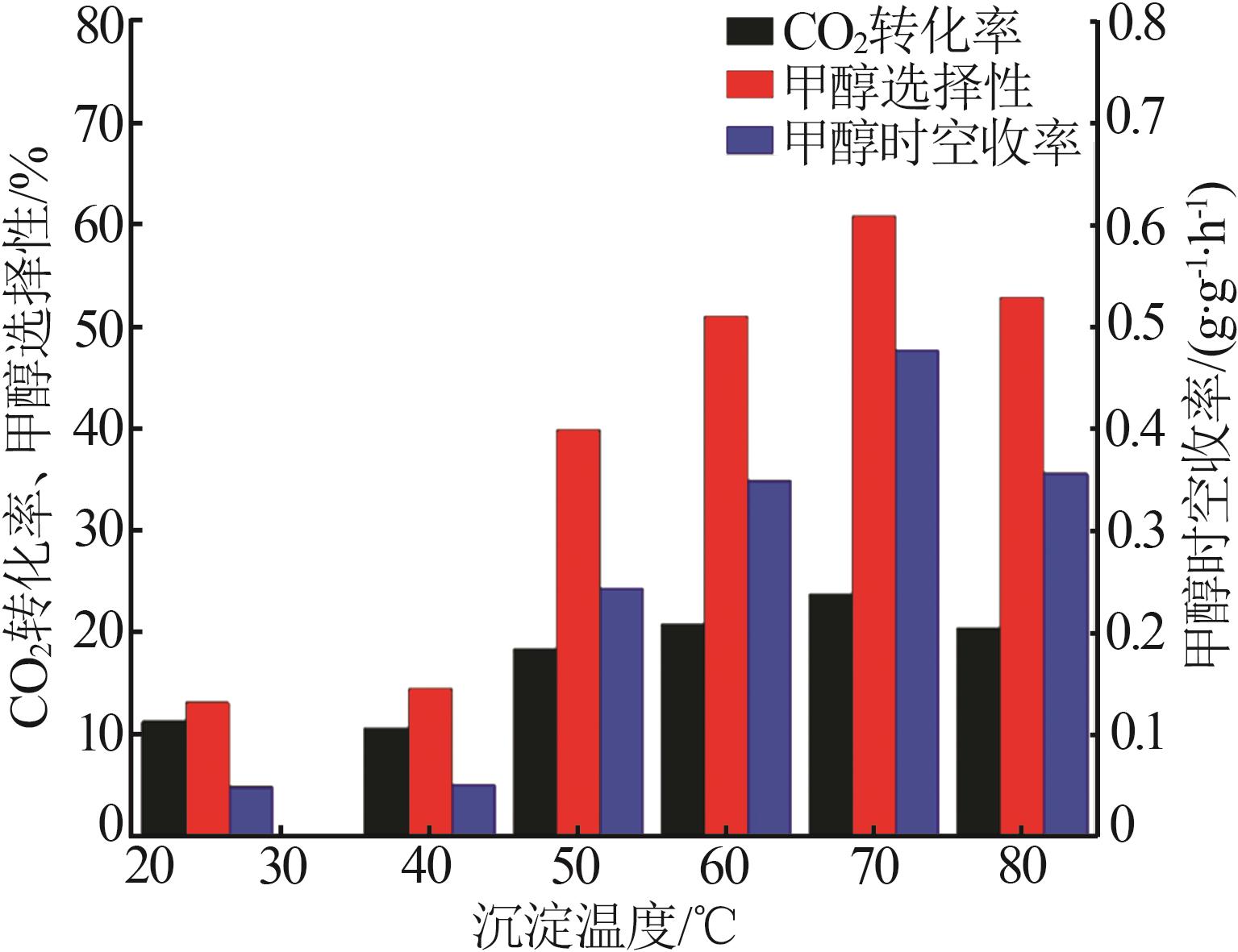
Inorganic Chemicals Industry ›› 2026, Vol. 58 ›› Issue (2): 117-124.doi: 10.19964/j.issn.1006-4990.2025-0005
• Catalytic Materials • Previous Articles
Study on effect of precipitation temperature on structure and catalytic performance of catalysts over hydrogenation of carbon dioxide to methanol
LI Tong( ), LI Nina, LI Yanpeng, WANG Xiaolong, WANG Panpan, WU Ping, ZHOU Yunyun, ZHU Chuanzong
), LI Nina, LI Yanpeng, WANG Xiaolong, WANG Panpan, WU Ping, ZHOU Yunyun, ZHU Chuanzong
- Zhejiang Green Intelligent Vehicle and Components Technology Innovation Center,Ningbo 315000,China
-
Received:2025-01-02Online:2026-02-10Published:2026-03-12
CLC Number:
Cite this article
LI Tong, LI Nina, LI Yanpeng, WANG Xiaolong, WANG Panpan, WU Ping, ZHOU Yunyun, ZHU Chuanzong. Study on effect of precipitation temperature on structure and catalytic performance of catalysts over hydrogenation of carbon dioxide to methanol[J]. Inorganic Chemicals Industry, 2026, 58(2): 117-124.
share this article
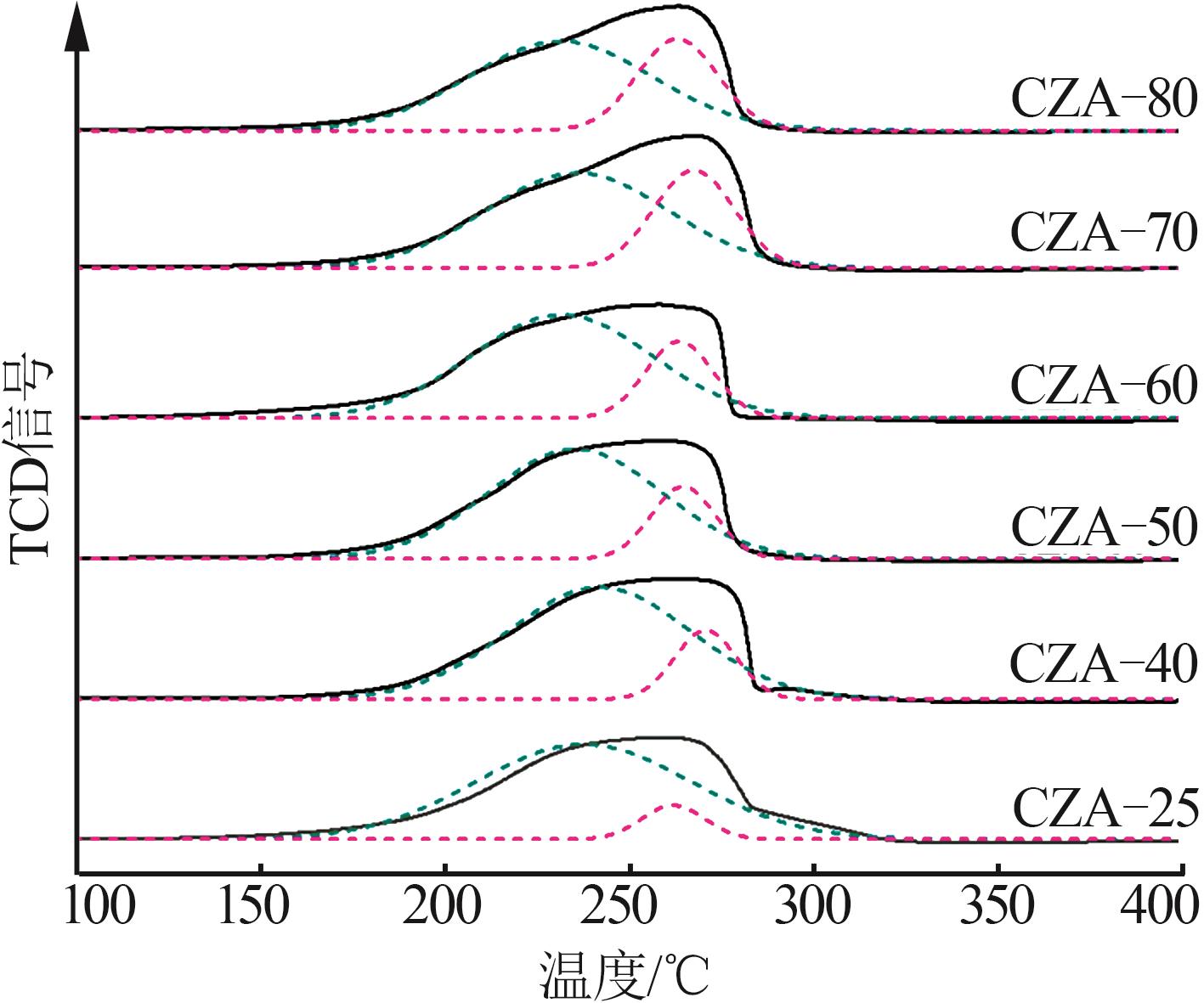
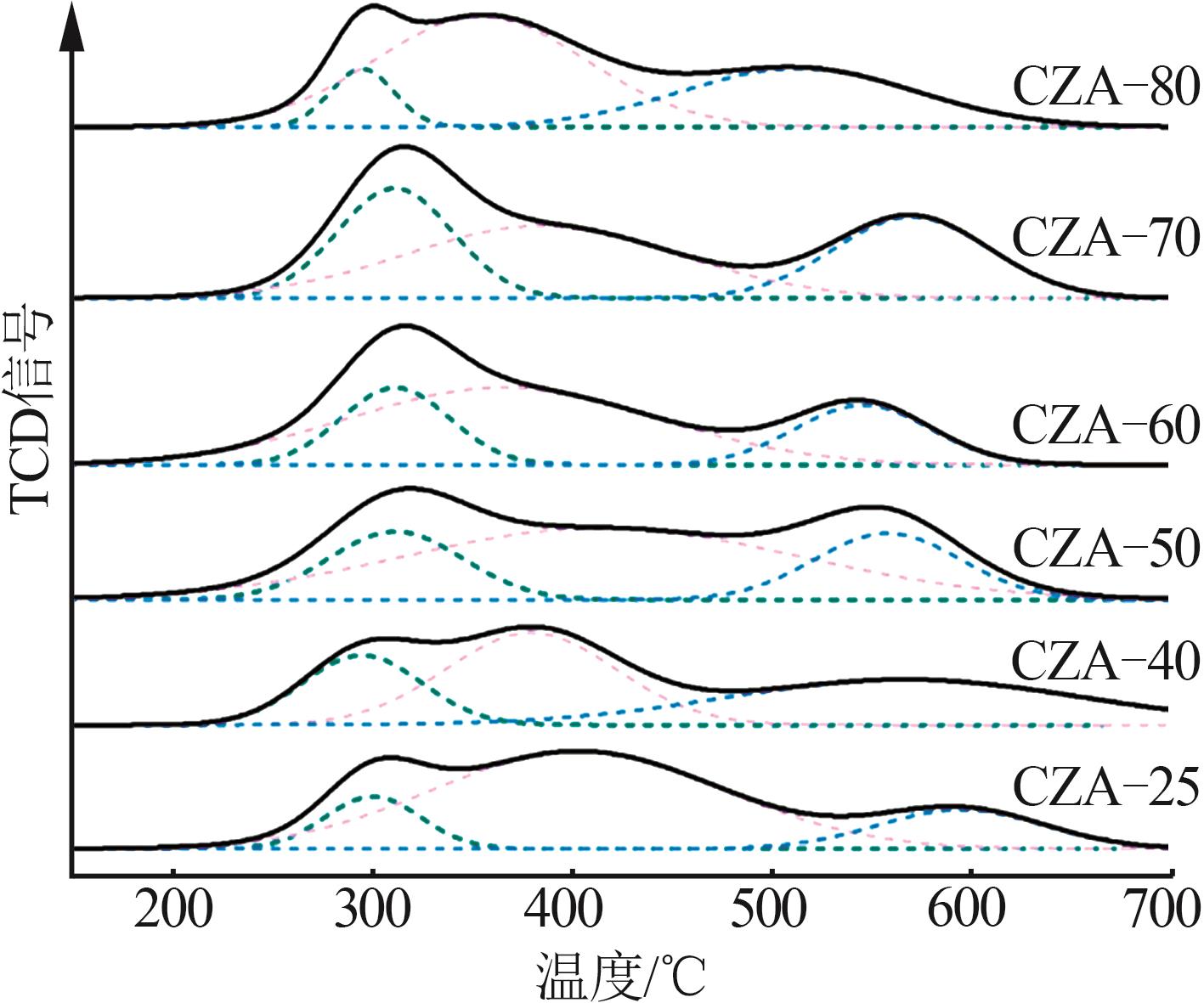
Table 1
Analysis of elements content and pore structure properties of catalysts after calcination"
| 催化剂 | w(元素组成)/% | 比表面积/ (m2·g-1) | 孔容/ (cm3·g-1) | 平均孔径/nm | ||
|---|---|---|---|---|---|---|
| CuO | ZnO | Al2O3 | ||||
| CZA-25 | 62.12 | 28.91 | 8.97 | 37.9 | 0.106 | 10.4 |
| CZA-40 | 62.28 | 29.07 | 8.65 | 42.5 | 0.131 | 8.4 |
| CZA-50 | 61.96 | 29.08 | 8.96 | 50.5 | 0.197 | 14.6 |
| CZA-60 | 62.03 | 28.90 | 9.07 | 52.2 | 0.180 | 12.5 |
| CZA-70 | 62.21 | 29.08 | 8.71 | 71.2 | 0.231 | 11.9 |
| CZA-80 | 62.13 | 28.92 | 8.95 | 49.9 | 0.130 | 9.9 |
| [1] | JIANG Xiao, NIE Xiaowa, GUO Xinwen,et al.Recent advances in carbon dioxide hydrogenation to methanol via heterogeneous catalysis[J].Chemical Reviews,2020,120(15):7984-8034. |
| [2] | YU Minli, WANG Ke, VREDENBURG H.Insights into low-carbon hydrogen production methods:Green,blue and aqua hydrogen[J].International Journal of Hydrogen Energy,2021,46(41):21261-21273. |
| [3] | NAVARRO-JAÉN S, VIRGINIE M, BONIN J,et al.Highlights and challenges in the selective reduction of carbon dioxide to methanol[J].Nature Reviews Chemistry,2021,5(8):564-579. |
| [4] | 张培培,王文波,米晓彤,等.ZnO/ZrO2固溶体催化剂的合成及其在CO2加氢制甲醇中的应用研究[J].无机盐工业,2025,57(5):125-132. |
| ZHANG Peipei, WANG Wenbo, MI Xiaotong,et al.Synthesis of ZnO/ZrO2 solid solution catalyst and its application in CO2 hydrogenation to methanol[J].Inorganic Chemicals Industry,2025,57(5):125-132. | |
| [5] | 占思进,刘仕轲,刘飞,等.ZnO-CeO2制备及催化性能研究[J].无机盐工业,2024,56(3):137-143. |
| ZHAN Sijin, LIU Shike, LIU Fei,et al.Study on preparation and catalytic performance of ZnO-CeO2 [J].Inorganic Chemicals Industry,2024,56(3):137-143. | |
| [6] | 任启霞,杨坤,刘飞,等.助剂对ZnO/ZrO2物化性质及催化性能的影响[J].无机盐工业,2024,56(3):144-154. |
| REN Qixia, YANG Kun, LIU Fei,et al.Effect of promoter on physicochemical properties and catalytic performance of ZnO/ZrO2 [J].Inorganic Chemicals Industry,2024,56(3):144-154. | |
| [7] | 胡博,王健捷,肖霞,等.二氧化碳加氢制甲醇催化剂研究进展[J].化学通报,2024,87(6):685-692. |
| HU Bo, WANG Jianjie, XIAO Xia,et al.Recent progress in catalysts for the hydrogenation of carbon dioxide to methanol[J].Che-mistry,2024,87(6):685-692. | |
| [8] | ARANDIA A,YIM J, WARRAICH H,et al.Effect of atomic layer deposited zinc promoter on the activity of copper-on-zirconia catalysts in the hydrogenation of carbon dioxide to methanol[J].Applied Catalysis B:Environmental,2023,321:122046. |
| [9] | WANG Shuai, SONG Lixin, QU Zhenping.Cu/ZnAl2O4 catalysts prepared by ammonia evaporation method:Improving methanol selectivity in CO2 hydrogenation via regulation of metal-support interaction[J].Chemical Engineering Journal,2023,469:144008. |
| [10] | 姜峰,徐云强,徐艳,等.铜锌铝催化剂催化脂肪酸甲酯加氢反应研究[J].化学工业与工程,2024,41(5):154-163. |
| JIANG Feng, XU Yunqiang, XU Yan,et al.Study on hydrogenation of fatty acid methyl ester catalyzed by Cu-Zn-Al catalyst[J].Chemical Industry and Engineering,2024,41(5):154-163. | |
| [11] | SUHAIL Z, KOCH C J, GOEPPERT A,et al.Integrated carbon dioxide capture and conversion to methanol utilizing tertiary amin-es over a heterogenous Cu/ZnO/Al2O3 catalyst[J].Langmuir,2024,40(10):5401-5408. |
| [12] | 林胜达,唐浩东,吕兆坡,等.沉淀方法对铜基甲醇合成催化剂前驱体及其性能的影响[J].催化学报,2010,31(10):1257-1262. |
| LIN Shengda, TANG Haodong, Zhaopo LÜ,et al.Influence of precipitation methods on precursors and properties of Cu-based catalyst for methanol synthesis[J].Chinese Journal of Catalysis,2010,31(10):1257-1262. | |
| [13] | HOU Xiaoxiao, XU Chenghua, LIU Yulu,et al.Improved methanol synthesis from CO2 hydrogenation over CuZnAlZr catalysts with precursor pre-activation by formaldehyde[J].Journal of Catalysis,2019,379:147-153. |
| [14] | 任超,徐波,王安杰,等.铜锌铝催化剂制备方法对CO2加氢反应性能影响[J].洁净煤技术,2022,28(1):70-76. |
| REN Chao, XU Bo, WANG Anjie,et al.Influence of the preparation method of copper-zinc-aluminum catalyst on the performance of carbon dioxide CO2 hydrogenation reaction[J].Clean Coal Technology,2022,28(1):70-76. | |
| [15] | VAFAEE ZONOUZ H, BARZEGARI F, REZAEI M.Development of CuO/ZnO/Al2O3-hydrotalcite-based catalysts for middle temperature water gas shift reaction:Impact of calcination temperature and residual carbonates[J].Fuel,2024,378:132882. |
| [16] | 王丹君,陶芙蓉,赵华华,等. Cu/ZnO/Al2O3催化剂的共沉淀-蒸氨法制备及其对二氧化碳加氢制甲醇的研究[J].分子催化,2011,25(2):124-129. |
| WANG Danjun, TAO Furong, ZHAO Huahua,et al.Preparation of Cu/ZnO/Al2O3 catalyst via coprecipition-amonia evaporation for methanol synthesis from CO2 and H2 [J].Journal of Molecular Catalysis,2011,25(2):124-129. | |
| [17] | LI J L, INUI T.Characterization of precursors of methanol synthesis catalysts,copper/zinc/aluminum oxides,precipitated at different pHs and temperatures[J].Applied Catalysis A:General,1996,137(1):105-117. |
| [18] | 沈百荣,方志刚,孙琦,等.EXAFS研究合成甲醇催化剂 Cu/ZnO/Al2O3的制备条件[J].复旦学报(自然科学版),1999,38(6):681-684. |
| SHEN Bairong, FANG Zhigang, SUN Qi,et al.EXAFS study on the preparation of Cu/ZnO/Al2O3 catalyst for methanol synthesis[J].Journal of Fudan University,1999,38(6):681-684. | |
| [19] | 姜秀云,杨文兵,宋昊,等.甲酸辅助Cu-ZnO-Al2O3催化剂制备及其CO2加氢制甲醇性能研究[J].燃料化学学报(中英文),2023,51(1):120-128. |
| JIANG Xiuyun, YANG Wenbing, SONG Hao,et al.Formic acid assisted synthesis of Cu-ZnO-Al2O3 catalyst and its performance in CO2 hydrogenation to methanol[J].Journal of Fuel Chemistry and Technology,2023,51(1):120-128. | |
| [20] | 孔秀琴,唐兴江,许珊,等.溶胶-凝胶自燃烧法制备的CuO-ZnO/Al2O3及催化二氧化碳加氢制甲醇的性能研究[J].分子催化,2013,27(2):159-165. |
| KONG Xiuqin, TANG Xingjiang, XU Shan,et al.Preparation of CuO-ZnO/Al2O3 by sol-gel auto-combustion method and its catalytic property for methanol synthesis from CO2 hydrogenation[J].Journal of Molecular Catalysis,2013,27(2):159-165. | |
| [21] | 张维中,温月丽,宋镕鹏,等.催化剂表面Cu0含量对二氧化碳加氢合成C2+醇性能的影响[J].高等学校化学学报,2020,41(6):1297-1305. |
| ZHANG Weizhong, WEN Yueli, SONG Rongpeng,et al.Effect of Cu0 content on catalyst surface on the performance of hydrogenation of carbon dioxide to C2+ alcohol[J].Chemical Journal of Chinese Universities,2020,41(6):1297-1305. | |
| [22] | LIU Haoran, YU Zhiqing, HUANG Wenbin,et al.Effect of Ce modification on the performance of CuLDH catalyst for CO2 hydrogenation to methanol[J].Journal of Fuel Chemistry and Technology,2024,52(2):159-170. |
| [23] | 刘华伟,薛静丽,钱胜涛,等.铜锌铝系醋酸乙酯加氢制乙醇催化剂的性能研究[J].天然气化工(C1化学与化工),2021,46(3):53-56,64. |
| LIU Huawei, XUE Jingli, QIAN Shengtao,et al.Study on performance of Cu-Zn-Al based catalyst for hydrogenation of ethyl acetate to ethanol[J].Natural Gas Chemical Industry,2021,46(3):53-56,64. | |
| [24] | SUN Jiyu, LIU Fangyuan, SALAHUDDIN U,et al.Optimization and understanding of ZnO nanoarray supported Cu-ZnO-Al2O3 catalyst for enhanced CO2-methanol conversion at low temperature and pressure[J].Chemical Engineering Journal,2023,455:140559. |
| [25] | 张兰,陈标华,王宁.CuZn/CeO2催化剂在CO2加氢制甲醇中的应用研究[J].低碳化学与化工,2024,49(8):100-106. |
| ZHANG Lan, CHEN Biaohua, WANG Ning.Study on application of CuZn/CeO2 catalysts in CO2 hydrogenation to methanol[J].Low-Carbon Chemistry and Chemical Engineering,2024,49(8):100-106. | |
| [26] | LI Huixiang, NIE Xuezhong, DU Hong,et al.Understanding the role of base species on reversed Cu catalyst in ring opening of furan compounds to 1,2-pentanediol[J].ChemSusChem,2024,17(1):e202300880. |
| [27] | 杨强,王刚,李春山.Cu基催化剂表面改性及其催化二氧化碳加氢制甲醇性能研究[J].过程工程学报,2024,24(10):1166-1176. |
| YANG Qiang, WANG Gang, LI Chunshan.Surface modification and catalytic performance study of Cu-based carbon dioxide to methanol hydrogenation catalyst[J].The Chinese Journal of Process Engineering,2024,24(10):1166-1176. | |
| [28] | 黄浩,纳薇,高文桂,等.La-Cu/ZnO/ZrO2催化剂上CO2加氢合成甲醇性能研究[J].功能材料,2024,55(9):9087-9093. |
| HUANG Hao, NA Wei, GAO Wengui,et al.Study on the performance of CO2 hydrogenation to methanol on La-Cu/ZnO/ZrO2 catalysts[J].Journal of Functional Materials,2024,55(9):9087-9093. | |
| [29] | ZHANG Yanfei, ZHONG Liangshu, WANG Hui,et al.Catalytic performance of spray-dried Cu/ZnO/Al2O3/ZrO2 catalysts for slurry methanol synthesis from CO2 hydrogenation[J].Journal of CO2 Utilization,2016,15:72-82. |
| [30] | CHEN Shuyao, ZHANG Junfeng, WANG Peng,et al.Effect of vapor-phase-treatment to CuZnZr catalyst on the reaction behaviors in CO2 hydrogenation into methanol[J].ChemCatChem,2019,11(5):1448-1457. |
| [1] | ZHANG Peipei, WANG Wenbo, MI Xiaotong, XIN Jing, LI Yongheng, LI Sixuan, XIA Lin. Study on synthesis of ZnO/ZrO2 catalyst and its application in CO2 hydrogenation to methanol [J]. Inorganic Chemicals Industry, 2025, 57(5): 125-132. |
| [2] | KANG Jianming, HAN Xushen, JIN Yan, YU Jianguo. Study on biological contact oxidation treatment technology of ultra-hypersaline methanol-containing gas field produced water [J]. Inorganic Chemicals Industry, 2025, 57(10): 98-102. |
| [3] | LI Yongheng, MI Xiaotong, ZHANG Peipei. Synthesis of holycrystalline nano-ZSM-5 aggregate and its application in methylation of toluene [J]. Inorganic Chemicals Industry, 2024, 56(5): 135-140. |
| [4] | ZHAN Sijin, LIU Shike, LIU Fei, YAO Mengqin, CAO Jianxin. Study on preparation and catalytic performance of ZnO-CeO2 [J]. Inorganic Chemicals Industry, 2024, 56(3): 137-143. |
| [5] | REN Qixia, YANG Kun, LIU Fei, YAO Mengqin, CAO Jianxin. Effect of promoter on physicochemical properties and catalytic performance of ZnO/ZrO2 [J]. Inorganic Chemicals Industry, 2024, 56(3): 144-154. |
| [6] | WANG Zihan, LI Jun, CHEN Ming, ZHOU Qingyu. Study on preparation of battery grade ferric phosphate by co-precipitation of ferric nitrate and phosphoric acid [J]. Inorganic Chemicals Industry, 2023, 55(7): 51-57. |
| [7] | LI He, ZHANG Lijie, ZHANG Kai, SU Jin, YAO Zhaoyang, ZENG Xianjun, GUO Chunlei, SUN Yanmin. Advances in technology and catalyst for methanol oxidized to formaldehyde [J]. Inorganic Chemicals Industry, 2023, 55(11): 12-18. |
| [8] | YANG Tinglong,WANG Fuzhong,LIU Fei. Study on sulfur poisoning of zirconium-based bimetallic oxides catalyst [J]. Inorganic Chemicals Industry, 2023, 55(1): 151-158. |
| [9] | JI Chao,WU Luming,LI Bin,ZANG Jiazhong,YU Haibin. Research progress on modification of SAPO-34 as catalyst for methanol to olefins [J]. Inorganic Chemicals Industry, 2022, 54(7): 1-9. |
| [10] | Peng Xiaowei,Wang Yinbin,Zang Jiazhong,Yu Haibin. Research on preparation of metal modified catalysts and catalytic performances of methanol aromatization [J]. Inorganic Chemicals Industry, 2021, 53(9): 104-108. |
| [11] | Li Xiaoguo, Li Yongheng, Hou Zhanggui, Han Guodong, Xiao Jiawang, Gao Jinping, Chang Yang. Study on pilot catalytic performance of the modified catalyst for alkylation of toluene with methanol to p-xylene [J]. Inorganic Chemicals Industry, 2021, 53(3): 97-101. |
| [12] | Hou Zhanggui,Zhu Qianqian,Li Xiaoguo,Li Yongheng,Chang Yang,Zhang Anfeng,Guo Xinwen. Preparation of modificated micro-mesoporous ZSM-5 molecular sieve catalyst and its catalytic performance for alkylation of toluene with methanol [J]. Inorganic Chemicals Industry, 2020, 52(9): 96-99. |
| [13] | Su Wei,Zhang Linfeng,Lü Qingyang,Xia Yuyan,Yuan Hua. Synthesis of diphenyl carbonate by oxidative carbonylation over palladium loaded MnOx-based bimetallic oxide catalysis [J]. Inorganic Chemicals Industry, 2019, 51(9): 91-96. |
| [14] | Liu Zhikai1,Cao Hui2,Wang Tianyun1,Han Guodong2,Liu Yi1,Zhang Yi1. Study on novel catalysts for direct catalytic conversion of unconventional natural gas to aromatics [J]. Inorganic Chemicals Industry, 2019, 51(12): 83-88. |
| [15] | Li Shuwen,Zhou Yan,Wang Tielin. Study on preparation and photocatalysis reduction for CO2 of BiVO4/rGO composite [J]. Inorganic Chemicals Industry, 2019, 51(11): 82-87. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||
|
||