Inorganic Chemicals Industry ›› 2026, Vol. 58 ›› Issue (3): 85-95.doi: 10.19964/j.issn.1006-4990.2025-0113
• Environment·Health·Safety • Previous Articles Next Articles
Study on preparation of electrolytic manganese slag-based composites and their Cd2+ removal
HAN Qingwen1( ), YUAN Jiaxin2,3, ZHAN Wei2,3(
), YUAN Jiaxin2,3, ZHAN Wei2,3( ), HUANG Ping2,3, NAN Fangming2,3, CHEN Donghui2,3, TAN Jie2,3
), HUANG Ping2,3, NAN Fangming2,3, CHEN Donghui2,3, TAN Jie2,3
- 1.Hubei Three Gorges Laboratory,Yichang 443007,China
2.Hubei Engineering Research Center for Heavy Metal Pollution Prevention and Control,School of Resources and Environment,South-Central Minzu University,Wuhan 430074,China
3.Key Laboratory of Catalysis and Energy Materials Chemistry of Ministry of Education,Wuhan 430074,China
-
Received:2025-03-08Online:2026-03-10Published:2025-09-25 -
Contact:ZHAN Wei E-mail:qingwenhan@qq.com;zw7789818@mail.scuec.edu.cn
CLC Number:
Cite this article
HAN Qingwen, YUAN Jiaxin, ZHAN Wei, HUANG Ping, NAN Fangming, CHEN Donghui, TAN Jie. Study on preparation of electrolytic manganese slag-based composites and their Cd2+ removal[J]. Inorganic Chemicals Industry, 2026, 58(3): 85-95.
share this article
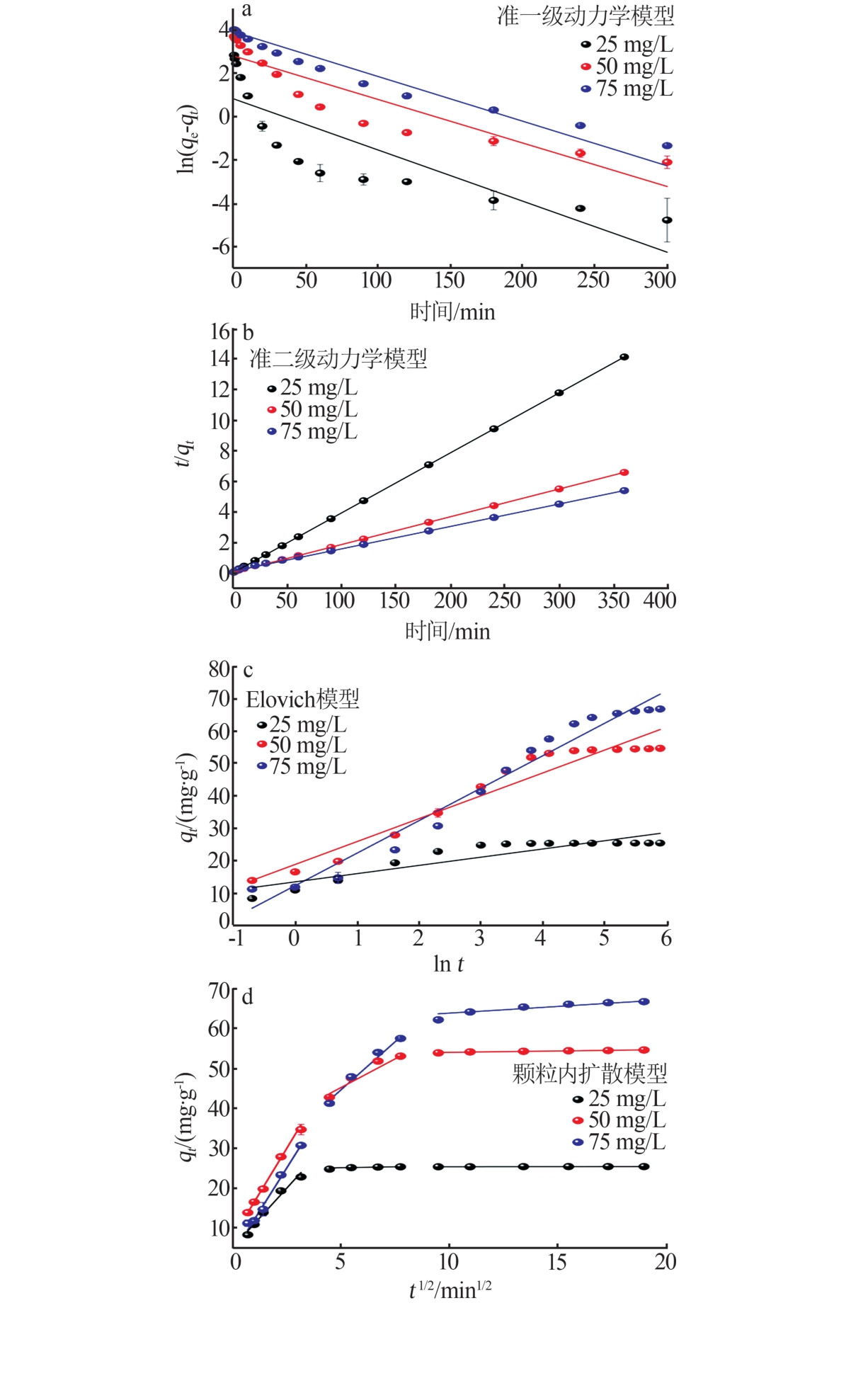
Table 2
Kinetic model rate constants of adsorption"
质量浓度/ (mg·L-1) | 准一级动力学模型 | 准二级动力学模型 | Elovich模型 | ||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
qe/ (mg·g-1) | k1/ h-1 | R2 | qe/ (mg·g-1) | k2/ (g·mg-1·h-1) | R2 | β/ (mg·g-1) | α/ (mg·g-1·min-1) | R2 | |||
| 25 | 2.28 | 1.42 | 0.684 9 | 25.48 | 6.40 | 1.000 0 | 0.39 | 3.07 | 0.811 4 | ||
| 50 | 16.56 | 1.21 | 0.829 1 | 55.04 | 0.47 | 0.999 9 | 0.14 | 10.31 | 0.935 4 | ||
| 75 | 50.48 | 1.24 | 0.945 5 | 68.12 | 0.12 | 0.999 8 | 0.10 | 22.74 | 0.966 0 | ||
Table 3
Fitting parameters of dynamics model of intra-particle diffusion"
质量浓度/ (mg·L-1) | 表面扩散阶段 | 孔扩散阶段 | 平衡阶段 | ||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
qt / (mg·g-1) | kp,1/ (mg·g-1·min-1/2) | R2 | qt / (mg·g-1) | kp,2/ (mg·g-1·min-1/2) | R2 | C/ (mg·g-1) | kp,3/ (mg·g-1·min-1/2) | R2 | |||
| 25 | 60.56 | 0.34 | 0.843 6 | 20.55 | 4.83 | 0.977 9 | 3.45 | 8.65 | 0.991 0 | ||
| 50 | 53.41 | 0.07 | 0.890 9 | 30.87 | 2.88 | 0.976 4 | 8.97 | 7.57 | 0.997 6 | ||
| 75 | 25.34 | 0.01 | 0.929 8 | 24.67 | 0.10 | 0.636 0 | 5.07 | 5.98 | 0.968 6 | ||
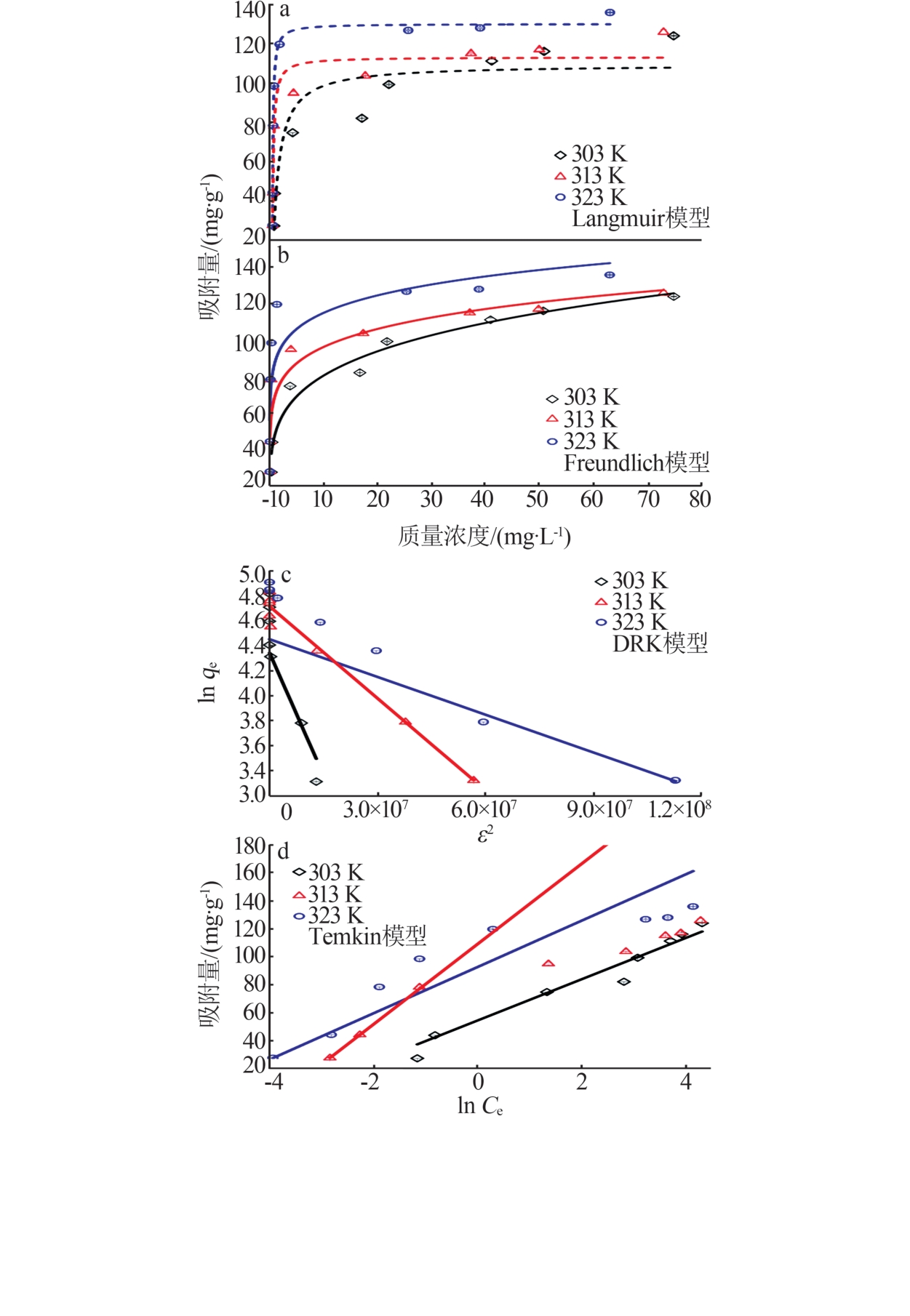
Table 4
Fitting parameters of adsorption isotherm model"
温度/ K | Langmuir | Freundlich | DRK | Temkin | |||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
qe/ (mg·g-1) | KL/ (L·mg-1) | R2 | 1/n | KF/[(mg·g-1)· (L·mg-1)1/n ] | R2 | E | qm/ (mg·g-1) | R2 | B1 | Kt / (L·mol-1) | R2 | ||||
| 303 | 109.20 | 0.96 | 0.852 5 | 0.22 | 48.32 | 0.959 9 | 2 761.67 | 77.03 | 0.973 6 | 54.36 | 1.31 | 0.952 1 | |||
| 313 | 113.05 | 5.99 | 0.939 6 | 0.14 | 69.44 | 0.885 3 | 4 506.94 | 111.85 | 0.999 6 | 73.60 | 1.18 | 0.931 1 | |||
| 323 | 129.96 | 9.77 | 0.989 1 | 0.12 | 87.10 | 0.745 2 | 5 918.22 | 123.98 | 0.971 9 | 92.57 | 1.20 | 0.936 6 | |||
| [1] | BHATTACHARYYA K,SEN D, BANIK A K,et al.Adsorptive removal of cadmium from aqueous medium-a critical review[J].Phy-sics and Chemistry of the Earth, A/B/C Parts,2024,134:103538. |
| [2] | 兰馨,郭帅庭,李彬.吸附法处理水中重金属的解吸附研究进展[J].山东化工,2020,49(10):81-82,84. |
| LAN Xin, GUO Shuaiting, LI Bin.Reviews on desorption of heavy metal in water by adsorption method[J].Shandong Chemical Industry,2020,49(10):81-82,84. | |
| [3] | 董馨予,王海峰,贺跃,等.电解锰渣的浸出毒性分析及无害化处理[J].无机盐工业,2023,55(5):85-90. |
| DONG Xinyu, WANG Haifeng, HE Yue,et al.Exleaching toxicity analysis and harmless treatment of electrolytic manganese resi-due[J].Inorganic Chemicals Industry,2023,55(5):85-90. | |
| [4] | HE Dejun, SHU Jiancheng, WANG Rong,et al.A critical review on approaches for electrolytic manganese residue treatment and disposal technology:Reduction,pretreatment,and reuse[J].Journal of Hazardous Materials,2021,418:126235. |
| [5] | 李家乐,万廷勇,柯平超,等.电解锰渣无害化与资源化处理技术研究进展[J].无机盐工业,2025,57(12):Doi:10.19964/j.issn.1006-4990.2025-0126. |
| LI Jiale, WAN Tingyong, KE Pingchao,et al.Research progress on harmless and resource treatment technology of electrolytic manganese residue[J].Inorganic Chemicals Industry,2025,57(12):Doi:10.19964/j.issn.1006-4990.2025-0126. | |
| [6] | 熊鑫,谢更新,晏铭,等.黏土矿物复合材料固化电解锰渣中Mn的研究[J].环境科学与技术,2022,45(9):57-63. |
| XIONG Xin, XIE Gengxin, YAN Ming,et al.Study on solidification efficiency of Mn in electrolytic manganese residue by clay mi-neral composites[J].Environmental Science & Technology,2022,45(9):57-63. | |
| [7] | 王鹏星,祝楚微,汪玉碧,等.电解锰渣有价元素回收及有害物质处理技术研究进展[J].化工矿物与加工,2022,51(7):1- 9. |
| WANG Pengxing, ZHU Chuwei, WANG Yubi,et al.Research progress of the technology for recovery of the valuable elements from electrolytic manganese slag and processing hazardous substances[J].Industrial Minerals & Processing,2022,51(7):1-9. | |
| [8] | FU Yong, QIAO Hongxia, FENG Qiong,et al.Review of new methods for resource utilisation of electrolytic manganese residue and its application in building materials[J].Construction and Building Materials,2023,401:132901. |
| [9] | SU Huili, ZHOU Wentao, Xianjun LYU,et al.Remediation treatment and resource utilization trends of electrolytic manganese residue[J].Minerals Engineering,2023,202:108264. |
| [10] | 孙煜琳,李杰瑞,苏向东,等.电解锰渣性质的研究进展及资源化利用展望[J].山东化工,2022,51(1):102-105. |
| SUN Yulin, LI Jierui, SU Xiangdong,et al.Research progress on properties on electrolytic manganese slag and prospect of resource utilization[J].Shandong Chemical Industry,2022,51(1):102-105. | |
| [11] | 唐雪莲,李会泉,胡应燕,等.电解锰渣资源化回收利用技术研究进展及“双碳”影响评估[J].有色金属(冶炼部分),2024(11):177-186. |
| TANG Xuelian, LI Huiquan, HU Yingyan,et al.Research progress on resource recovery and utilization of electrolytic manganese slag and impact assessment of carbon peak and carbon neutrality[J].Nonferrous Metals(Extractive Metallurgy),2024(11):177-186. | |
| [12] | HE Shichao, JIANG Daoyan, HONG Minghao,et al.Hazard-free treatment and resource utilisation of electrolytic manganese residue:A review[J].Journal of Cleaner Production,2021,306:127224. |
| [13] | 孙燕,蓝际荣,郭莉,等.利用电解锰渣制备As(Ⅲ)吸附材料及其性能研究[J].化工学报,2019,70(6):2377-2385. |
| SUN Yan, LAN Jirong, GUO Li,et al.Preparation of As(Ⅲ) adsorbent material by electrolytic manganese slag and its properti-es[J].CIESC Journal,2019,70(6):2377-2385. | |
| [14] | 马梦雨,魏华,宋小龙,等.电解锰渣-方解石复合吸附剂对水中镉的吸附[J].非金属矿,2020,43(3):89-92. |
| MA Mengyu, WEI Hua, SONG Xiaolong,et al.Adsorption behaviors of Cd(Ⅱ) by electrolytic manganese residues-calcite composite adsorbent[J].Non-Metallic Mines,2020,43(3):89-92. | |
| [15] | 马时成.利用锰渣制备吸附剂及对Cu(Ⅱ)的吸附研究 [D].贵阳:贵州师范大学,2020. |
| MA Shicheng.Preparation of absorbent based on manganese residue and adsorption of Cu(Ⅱ)[D].Guiyang:Guizhou normal University,2020. | |
| [16] | 魏华,宋小龙,杨秀端,等.电解锰渣-壳聚糖复合吸附剂对Cr(Ⅵ)的吸附[J].非金属矿,2021,44(6):101-104. |
| WEI Hua, SONG Xiaolong, YANG Xiuduan,et al.Adsorption behaviors of Cr(Ⅵ) by electrolytic manganese residues-chitosan adsorbent[J].Non-Metallic Mines,2021,44(6):101-104. | |
| [17] | TOPARE N S, WADGAONKAR V S.A review on application of low-cost adsorbents for heavy metals removal from wastewater[J].Materials Today:Proceedings,2023,77:8-18. |
| [18] | GAO Ming, WANG Wei, YANG Hongbing,et al.Efficient removal of fluoride from aqueous solutions using 3D flower-like hierarchical zinc-magnesium-aluminum ternary oxide microsphe-res[J].Chemical Engineering Journal,2020,380:122459. |
| [19] | MA Mengyu, DU Yaguang, BAO Shenxu,et al.Removal of cadmium and lead from aqueous solutions by thermal activated electrolytic manganese residues[J].Science of the Total Environment,2020,748:141490. |
| [20] | YIN Guangcai, CHEN Xingling, SARKAR B,et al.Co-adsorpt-ion mechanisms of Cd(Ⅱ) and As(Ⅲ) by an Fe-Mn binary oxide biochar in aqueous solution[J].Chemical Engineering Journal,2023,466:143199. |
| [21] | MA Mengyu, WANG Ting, KE Xuan,et al.A novel slag composite for the adsorption of heavy metals:Preparation,characterization and mechanisms[J].Environmental Research,2023,216:114442. |
| [22] | LAN Jirong, SUN Yan, GUO Li,et al.Highly efficient removal of As(Ⅴ) with modified electrolytic manganese residues(M-EMRs) as a novel adsorbent[J].Journal of Alloys and Compounds,2019,811:151973. |
| [23] | ZONG Yiming, WANG Xinxiang, ZHANG Hao,et al.Preparation of a ternary composite based on water caltrop shell derived biochar and gelatin/alginate for cadmium removal from contamina-ted water:Performances assessment and mechanism insight[J].International Journal of Biological Macromolecules,2023,234:123637. |
| [24] | JI Xianguo, LIU Yucan, GAO Zhonglu,et al.Efficiency and mechanism of adsorption for imidacloprid removal from water by Fe-Mg co-modified water hyacinth-based biochar:Batch adsorption,fixed-bed adsorption,and DFT calculation[J].Separation and Purification Technology,2024,330:125235. |
| [25] | BEHBAHANI E S, DASHTIAN K, GHAEDI M.Fe3O4-FeMoS4:Promise magnetite LDH-based adsorbent for simultaneous removal of Pb(Ⅱ),Cd(Ⅱ),and Cu(Ⅱ) heavy metal ions[J].Jo-urnal of Hazardous Materials,2021,410:124560. |
| [26] | SONG Wen, ZHANG Xue, ZHANG Lu,et al.Removal of various aqueous heavy metals by polyethylene glycol modified MgAl-LDH:Adsorption mechanisms and vital role of precipitation[J].Journal of Molecular Liquids,2023,375:121386. |
| [27] | HOSSAIN M T, KHANDAKER S, BASHAR M M,et al.Simultaneous toxic Cd(Ⅱ) and Pb(Ⅱ) encapsulation from contaminated water using Mg/Al-LDH composite materials[J].Journal of Molecular Liquids,2022,368:120810. |
| [28] | WANG Yifan, LI Jianen, XU Liang,et al.EDTA functionalized Mg/Al hydroxides modified biochar for Pb(Ⅱ) and Cd(Ⅱ) removal:Adsorption performance and mechanism[J].Separation and Purification Technology,2024,335:126199. |
| [29] | YIN Guangcai, SONG Xiaowang, TAO Lin,et al.Novel Fe-Mn binary oxide-biochar as an adsorbent for removing Cd(Ⅱ) from aqueous solutions[J].Chemical Engineering Journal,2020,389:124465. |
| [30] | AL-GHOUTI M A, DA’ANA D A.Guidelines for the use and interpretation of adsorption isotherm models:A review[J].Journal of Hazardous Materials,2020,393:122383. |
| [31] | XIAO Wei, LIAO Yumei, DENG Ling,et al.Simultaneous removal of arsenic and cadmium in aqueous solution by a novel hydrotalcite-like absorbent FeMnCa-LDHs[J].Inorganic Chemistry Communications,2024,163:112312. |
| [32] | NGUYEN H K D, VAN NGUYEN H, NGUYEN V A.Effect of synthetic conditions on the structure of mesoporous Mg-Al-Co hydrotalcite[J].Journal of Molecular Structure,2018,1171:25- 32. |
| [33] | XU Shaodong, LI Dong, GUO Xueyi,et al.Selenium(Ⅵ) removal from caustic solution by synthetic Ca-Al-Cl layered double hydroxides[J].Transactions of Nonferrous Metals Society of China,2019,29(8):1763-1775. |
| [34] | BIAN Liang, NIE Jianan, JIANG Xiaoqiang,et al.Selective adsorption of uranyl and potentially toxic metal ions at the core-shell MFe2O4-TiO2(M=Mn,Fe,Zn,Co,or Ni) nanoparticles[J].Journal of Hazardous Materials,2019,365:835-845. |
| [35] | LIU Bo, YUE Bo, HE Lili,et al.Synergistic solidification and mechanism research of electrolytic manganese residue and coal fly ash based on C-A-S-H gel material[J].Journal of Environmental Management,2024,365:121600. |
| [36] | ZHU Jin, BAIG S ALI, SHENG Tiantian,et al.Fe3O4 and MnO2 assembled on honeycomb briquette cinders(HBC) for arsenic removal from aqueous solutions[J].Journal of Hazardous Materials,2015,286:220-228. |
| [37] | LAN Jirong, SUN Yan, HUANG Ping,et al.Using Electrolytic Manganese Residue to prepare novel nanocomposite catalysts for efficient degradation of Azo Dyes in Fenton-like processes[J].Chemosphere,2020,252:126487. |
| [38] | DENG Jiahui, CHENG Yalin, LIU Min,et al.Fe-Mn co-embed carbon spheres for aqueous divalent Cd removal[J].Colloids and Surfaces A:Physicochemical and Engineering Aspects,2021,612:126013. |
| [39] | LI Mengke, HE Zhiguo, ZHONG Hui,et al.Highly efficient persulfate catalyst prepared from modified electrolytic manganese residues coupled with biochar for the roxarsone removal[J].Journal of Environmental Management,2023,328:116945. |
| [1] | LI Jiale, WAN Tingyong, KE Pingchao, ZHOU Yipeng, XU Lingling. Research progress on harmless and resource treatment technology of electrolytic manganese residue [J]. Inorganic Chemicals Industry, 2026, 58(3): 1-8. |
| [2] | LIANG Qingyang, XUE Fei, HUANG Xuquan, ZHAO Xiaorong, WANG Haojie, HAN Zilong, WANG Jun. Study on mechanism of preparation of porous ceramsite and fixation of heavy metal manganese from electrolytic manganese slag and multi-source solid waste [J]. Inorganic Chemicals Industry, 2025, 57(8): 102-109. |
| [3] | NI Dong, TANG Liang, HE Zhaoyi, WANG Jian, PEI Shanshan, XIA Lei. Study on sulfate activation performance of electrolytic manganese residue in hydrated lime-slag system [J]. Inorganic Chemicals Industry, 2024, 56(11): 151-157. |
| [4] | DONG Xinyu, WANG Haifeng, HE Yue, YANG Pan, WANG Song, YANG Chunyuan, WANG Qin, HUANG Bifang, WANG Jiawei. Exleaching toxicity analysis and harmless treatment of electrolytic manganese residue [J]. Inorganic Chemicals Industry, 2023, 55(5): 85-90. |
| [5] | Yang Xiaohong,Xue Xishi,Zhang Lulu,Chang Jun. Kinetics study of calcium leaching from electrolytic manganese residue by hydrochloric acid [J]. Inorganic Chemicals Industry, 2021, 53(1): 82-86. |
| [6] | Chang Jun, Jia Fukang, Hu Chengshan, Ye Qianxu. Adsorption of manganese ion by zeolite synthesized from electrolytic manganese residue [J]. Inorganic Chemicals Industry, 2019, 51(9): 61-66. |
| [7] | Zhou Hongyan,Chen Ping,Zhao Yanrong,Liu Rongjin,Wei Jiazhan. Sulfate activation of electrolytic manganese residue on heat-stewed steel slag activity [J]. Inorganic Chemicals Industry, 2019, 51(5): 66-69. |
| [8] | RAN Lan, LIU Shao-You- , WEN Zheng-Kang. Study on preparation of ceramic tiles with electrolytic manganese residue and waste glass admixture at low sintering temperature [J]. INORGANICCHEMICALSINDUSTRY, 2014, 46(7): 56-. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||
|
||