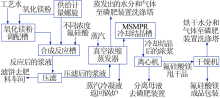
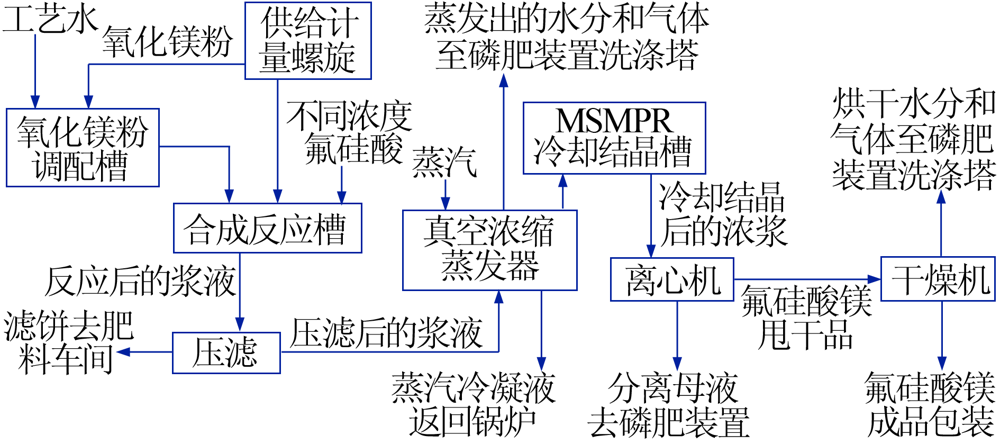
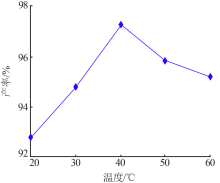
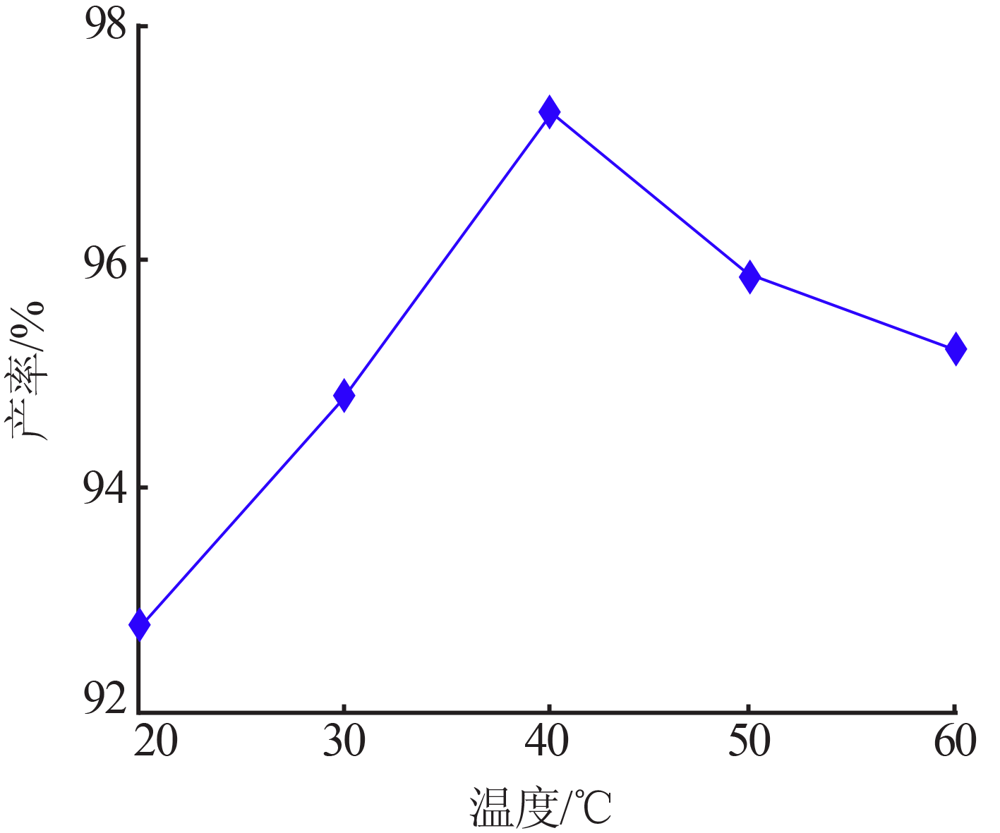
Inorganic Chemicals Industry ›› 2021, Vol. 53 ›› Issue (8): 79-82.doi: 10.19964/j.issn.1006-4990.2020-0523
• Industrial Techniques • Previous Articles Next Articles
Investigation on industrial production process of magnesium fluorosilicate
- Qinhuangdao Huaying Phosacid Co.,Ltd,Qinhuangdao 066004,China