Inorganic Chemicals Industry ›› 2023, Vol. 55 ›› Issue (12): 111-118.doi: 10.19964/j.issn.1006-4990.2023-0068
• Environment·Health·Safety • Previous Articles Next Articles
Occurrence and distribution of matter elements in phosphorus tailings
YANG Junjie1,2( ), DONG Yonggang2, ZHANG Jing1,2, ZHANG Yu1,2, CAO Jianxin1,3(
), DONG Yonggang2, ZHANG Jing1,2, ZHANG Yu1,2, CAO Jianxin1,3( )
)
- 1. School of Chemistry and Chemical Engineering,Guizhou University,Guiyang 550025,China
2. Guizhou Key Laboratory for Green Chemical and Clean Energy Technology,Guiyang 550025,China
3. Guizhou Provincial Engineering Research Center of Efficient Utilization for Waster,Guiyang 550025,China
-
Received:2023-02-13Online:2023-12-10Published:2023-12-14 -
Contact:CAO Jianxin E-mail:981961813@qq.com;jxcao@gzu.edu.cn
CLC Number:
Cite this article
YANG Junjie, DONG Yonggang, ZHANG Jing, ZHANG Yu, CAO Jianxin. Occurrence and distribution of matter elements in phosphorus tailings[J]. Inorganic Chemicals Industry, 2023, 55(12): 111-118.
share this article
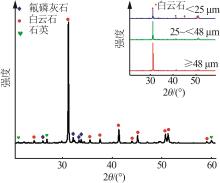
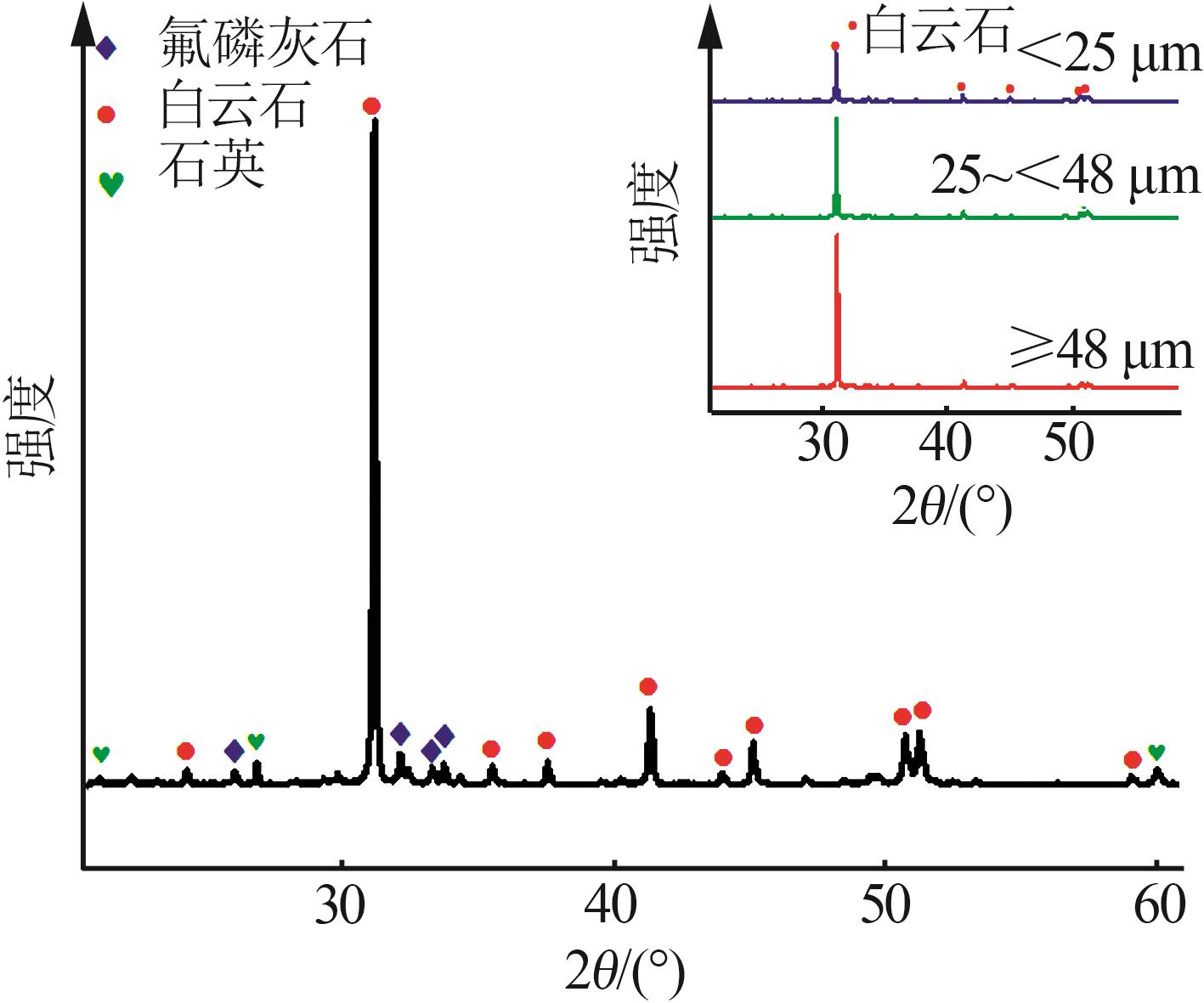
Table 2
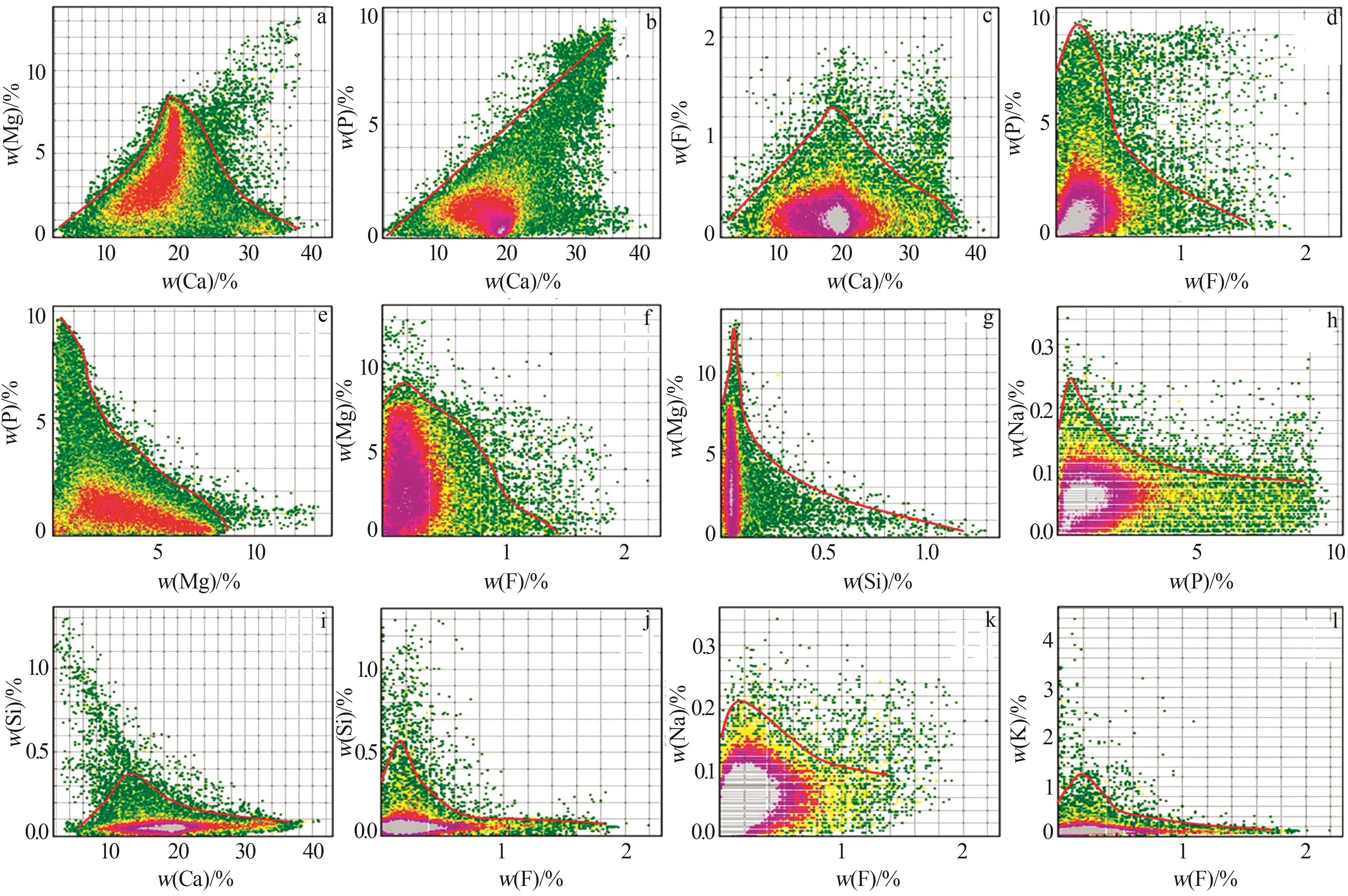
Mass fraction of major elements in phosphorus tailings with values of element transfer factor"
| 元素 | 磷尾矿 | 元素转移 因子/% | ||||
|---|---|---|---|---|---|---|
氧化物 形式 | 单元素 形式 | w(氧化 物)/% | w(单元素)/% | |||
| — | O | — | 36.829 | — | ||
| CaO | Ca | 45.674 | 29.711 | 34.147 | ||
| MgO | Mg | 17.661 | 10.651 | 97.038 | ||
| P2O5 | P | 6.424 | 2.713 | 7.917 | ||
| SiO2 | Si | 2.548 | 1.159 | 6.862 | ||
| F | F | 1.008 | 1.025 | 17.898 | ||
| Na2O | Na | 0.111 | 0.083 | 14.976 | ||
| K2O | K | 0.086 | 0.068 | 4.889 | ||
| Al2O3 | Al | 0.395 | 0.203 | 3.791 | ||
| SO3 | S | 0.987 | 0.378 | 95.281 | ||
| Fe2O3 | Fe | 0.483 | 0.289 | 7.399 | ||
| 其他 | 其他 | 0.123 | 0.091 | 9.318 | ||
| 烧失率 | 烧失率 | 24.500 | 16.800 | — | ||
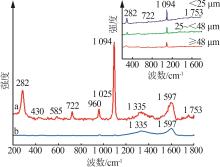
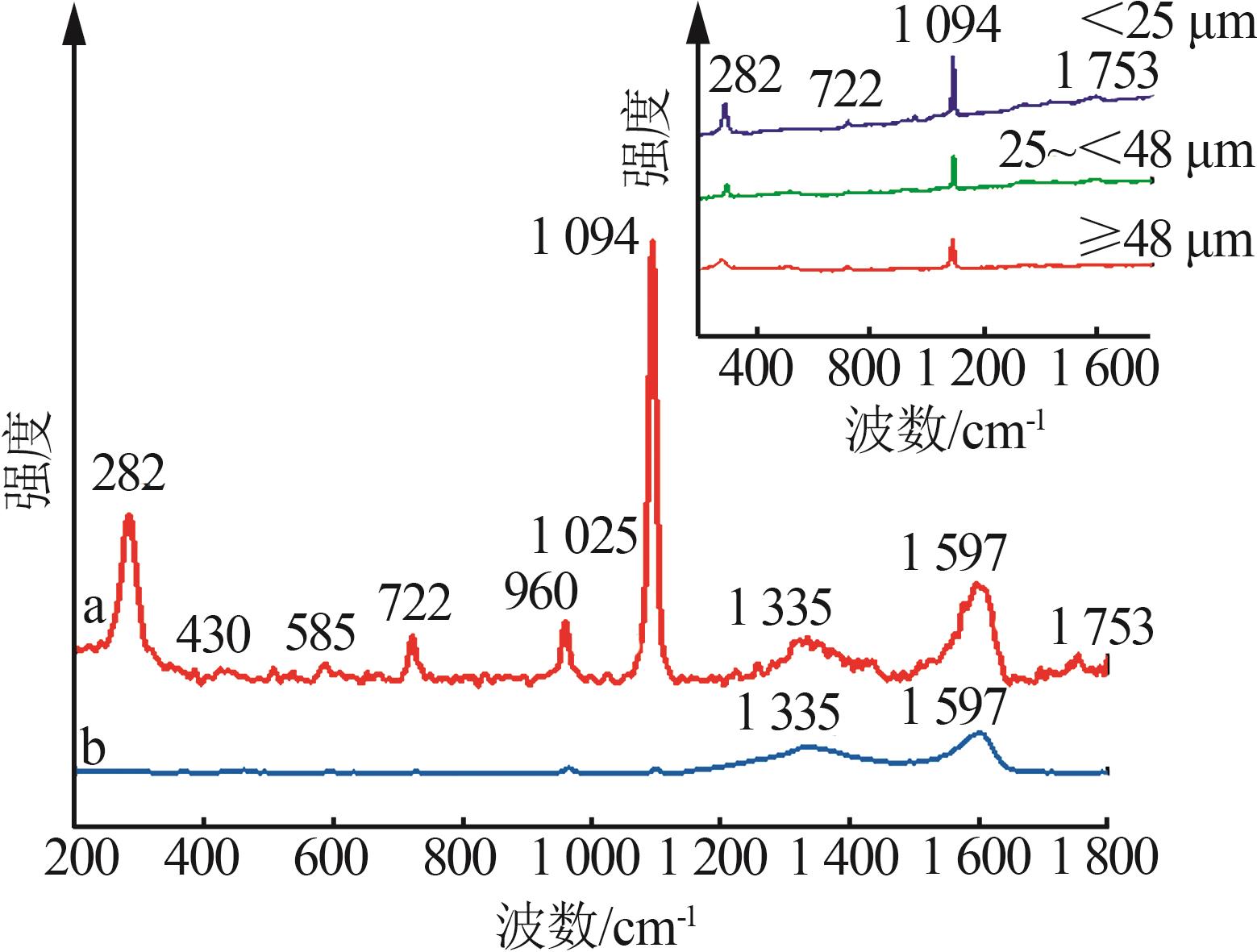
Table 4
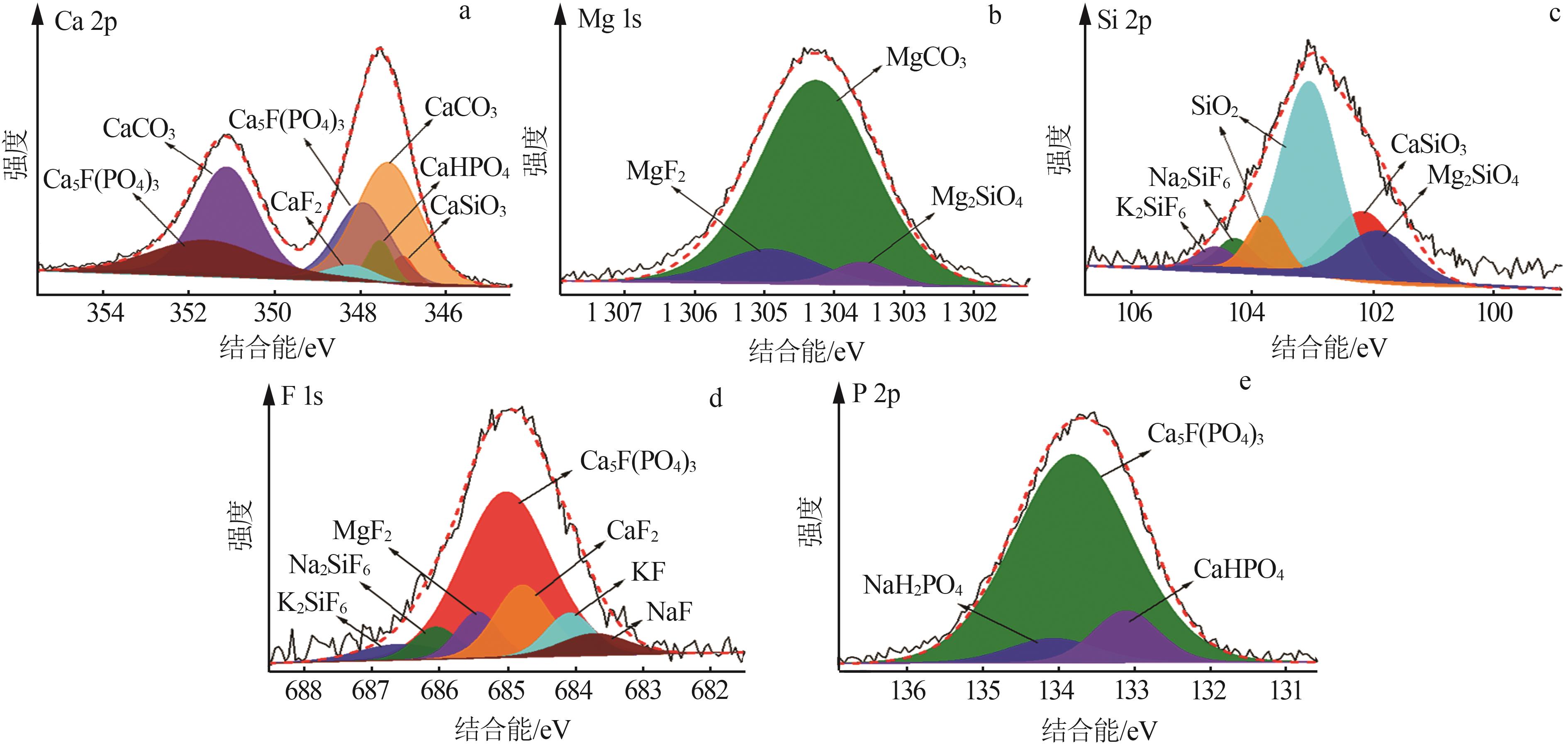
Phosphorus tailings surface composition corresponding to binding energy and relative content"
| 物质 | 结合能/eV | 相对含量/% | ||||
|---|---|---|---|---|---|---|
| Ca 2p | Mg 1s | Si 2p | F 1s | P 2p | ||
| CaCO3 | 347.30、351.10 | — | — | — | — | 32.52 |
| Ca5F(PO4)3 | 347.90、351.55 | — | — | 685.04 | 133.82 | 25.18 |
| CaF2 | 348.40 | — | — | 684.80 | — | 2.86 |
| CaSiO3 | 347.50 | — | 102.16 | — | — | 4.07 |
| CaHPO4 | 347.00 | — | — | — | 133.10 | 3.03 |
| MgF2 | — | 1 304.95 | — | 685.40 | — | 2.3 |
| MgCO3 | — | 1 034.25 | — | — | — | 17.53 |
| Mg2SiO4 | — | 1 303.60 | 101.90 | — | — | 2.76 |
| SiO2 | — | — | 103.00、103.80 | — | — | 6.21 |
| NaH2PO4 | — | — | — | — | 134.10 | 1.06 |
| Na2SiF6 | — | — | 104.30 | 686.00 | — | 0.86 |
| K2SiF6 | — | — | 104.60 | 686.60 | — | 0.70 |
| NaF | — | — | — | 683.70 | — | 0.39 |
| KF | — | — | — | 684.00 | — | 0.53 |
Table 5
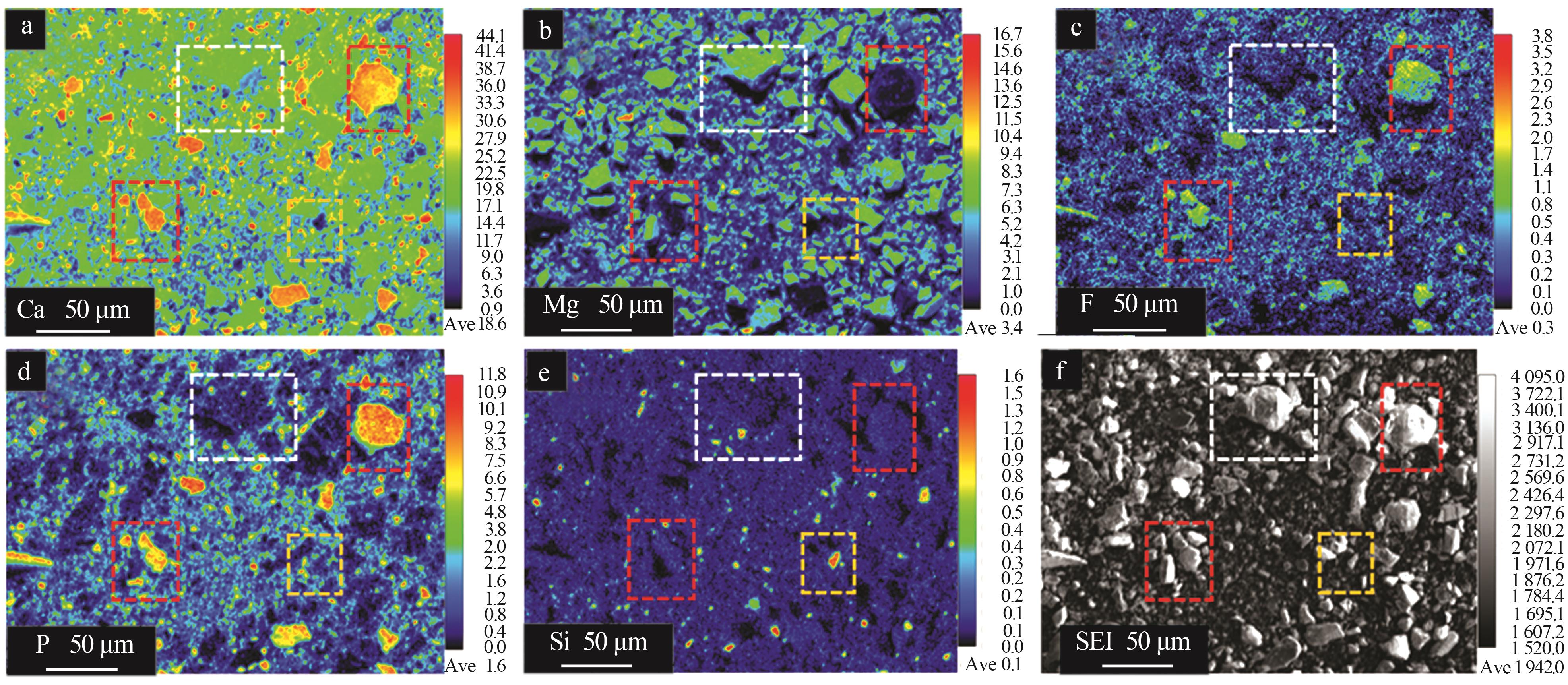
Corresponding EDS results of points displayed in Fig.6 %"
| 区域 | x(O) | x(Ca) | x(Mg) | x(P) | x(F) | x(Al) | x(Si) | x(Fe) | x(Na) | x(K) | x(S) |
|---|---|---|---|---|---|---|---|---|---|---|---|
| 1 | 64.79 | 19.16 | 15.04 | 0.22 | 0.13 | 0.01 | 0.06 | 0.14 | — | — | — |
| 2 | 68.54 | 2.17 | 2.52 | 0.77 | 0.33 | 0.66 | 24.39 | 0.09 | 0.06 | 0.19 | 0.27 |
| 3 | 65.60 | 16.43 | 1.98 | 9.65 | 4.89 | 0.13 | 0.69 | 0.03 | 0.22 | 0.02 | 0.36 |
| 4 | 70.70 | 11.86 | 6.73 | 5.46 | 4.72 | 0.23 | 0.13 | 0.01 | 0.05 | 0.01 | 0.11 |
| 1 | MAO Yuxuan, ZHANG Wen, LI Hongqiang, et al. Utilization of Fenton oxidation approach in wet phosphoric acid process for mitigating foam generation[J]. Colloids and Surfaces A:Physicochemical and Engineering Aspects, 2022, 648:129297. |
| 2 | 周雪娜, 关洪亮, 何东升, 等. 磷尾矿综合利用研究进展[J]. 广州化工, 2021, 49(5):24-27. |
| ZHOU Xuena, GUAN Hongliang, HE Dongsheng, et al. Research progress on comprehensive utilization by phosphate tailings[J]. Guangzhou Chemical Industry, 2021, 49(5):24-27. | |
| 3 |
CHEN Qiusong, ZHANG Qinli, FOURIE A, et al. Utilization of phosphogypsum and phosphate tailings for cemented paste back-fill[J]. Journal of Environmental Management, 2017, 201:19-27.
doi: S0301-4797(17)30608-4 pmid: 28633078 |
| 4 | 颜鑫, 魏义兰. 含钙镁废渣综合利用的现状及展望[J]. 无机盐工业, 2022, 54(1):7-11. |
| YAN Xin, WEI Yilan. Present situation and prospect of comprehensive utilization of waste residue containing calcium and magnesium[J]. Inorganic Chemicals Industry, 2022, 54(1):7-11. | |
| 5 | 张萍花, 燕云洁, 陈建钧, 等. 高镁钙磷尾矿酸解制纳米级碳酸钙[J]. 无机盐工业, 2019, 51(3):63-66. |
| ZHANG Pinghua, YAN Yunjie, CHEN Jianjun, et al. Preparation of nanometer calcium carbonate from high-Ca-Mg phosphorus tailings by acid leaching[J]. Inorganic Chemicals Industry, 2019, 51(3):63-66. | |
| 6 | MAO Song, ZHANG Qin. Mineralogical characteristics of phosphate tailings for comprehensive utilization[J]. Advances in Civil Engineering, 2021, 2021:1-13. |
| 7 |
PÉREZ-LÓPEZ R, NIETO J M, LÓPEZ-COTO I, et al. Dynamics of contaminants in phosphogypsum of the fertilizer industry of Huelva(SW Spain):From phosphate rock ore to the environ-ment[J]. Applied Geochemistry, 2010, 25(5):705-715.
doi: 10.1016/j.apgeochem.2010.02.003 |
| 8 |
YANG Xiaosheng, MAKKONEN H T, PAKKANEN L. Rare earth occurrences in streams of processing a phosphate ore[J]. Minerals, 2019, 9(5):262.
doi: 10.3390/min9050262 |
| 9 |
YANG Shuqi, YE Huijuan, LIU Yingxin. The different inclusions' characteristics between natural and heat-treated tanzanite:Evidence from Raman spectroscopy[J]. Crystals, 2021, 11(11):1302.
doi: 10.3390/cryst11111302 |
| 10 | 杜广鹏, 范建良. 方解石族矿物的拉曼光谱特征[J]. 矿物岩石, 2010, 30(4):32-35. |
| DU Guangpeng, FAN Jianliang. Characteristics of Raman spectral of calcite group minerals[J]. Journal of Mineralogy and Petrology, 2010, 30(4):32-35. | |
| 11 |
DUFRESNE W J B, RUFLEDT C J, MARSHALL C P. Raman spectroscopy of the eight natural carbonate minerals of calcite structure[J]. Journal of Raman Spectroscopy, 2018, 49(12):1999-2007.
doi: 10.1002/jrs.v49.12 |
| 12 |
DEMRI B, MUSTER D. XPS study of some calcium compounds[J]. Journal of Materials Processing Technology, 1995, 55(3/4):311-314.
doi: 10.1016/0924-0136(95)02023-3 |
| 13 | 孙伟, 唐鸿鹄, 陈臣. 萤石-白钨矿浮选分离体系中硅酸钠的溶液化学行为[J]. 中国有色金属学报, 2013, 23(8):2274-2283. |
| SUN Wei, TANG Honghu, CHEN Chen. Solution chemistry behavior of sodium silicate in flotation of fluorite and scheelite[J]. The Chinese Journal of Nonferrous Metals, 2013, 23(8):2274-2283. | |
| 14 |
MA Lei, HUANG Yi, ZHAO Kaixin, et al. Removal of uranium from acidic aqueous solution by natural fluorapatite[J]. Journal of Environmental Chemical Engineering, 2021, 9(6):106600.
doi: 10.1016/j.jece.2021.106600 |
| 15 | LAYROLLE P, LEBUGLE A. Synthesis in pure ethanol and characterization of nanosized calcium phosphate fluoroapatite[J]. Che- mistry of Materials, 1996, 8(1):134-144. |
| 16 |
SOSULNIKOV M I, TETERIN Y A. X-ray photoelectron studies of Ca,Sr and Ba and their oxides and carbonates[J]. Journal of Electron Spectroscopy and Related Phenomena, 1992, 59(2):111-126.
doi: 10.1016/0368-2048(92)85002-O |
| 17 |
LIU Weijun, ZHANG Jie, WANG Weiqing, et al. Flotation behaviors of ilmenite,titanaugite,and forsterite using sodium oleate as the collector[J]. Minerals Engineering, 2015, 72:1-9.
doi: 10.1016/j.mineng.2014.12.021 |
| 18 | 罗娜. 菱镁矿与白云石的浮选分离强化研究[D]. 沈阳: 东北大学, 2018. |
| LUO Na. Study on intensifying flotation separation of magnesite and dolomite[D]. Shenyang: Northeastern University, 2018. | |
| 19 |
DOMASHEVSKAYA E P, IVKOV S A, SITNIKOV A V, et al. The features of CoFeZr alloy nanocrystals formation in film composites of (CoFeZr) x (MgF2)100- x [J]. Journal of Alloys and Compounds, 2021, 870:159398.
doi: 10.1016/j.jallcom.2021.159398 |
| 20 |
MILLER M L, LINTON R W. X-ray photoelectron spectroscopy of thermally treated silica(SiO2) surfaces[J]. Analytical Chemistry, 1985, 57(12):2314-2319.
doi: 10.1021/ac00289a033 |
| 21 |
GROSS T, RAMM M, SONNTAG H, et al. An XPS analysis of different SiO2 modifications employing a C1 s as well as an Au4 f7/2 static charge reference[J]. Surface and Interface Analysis, 1992, 18(1):59-64.
doi: 10.1002/sia.v18:1 |
| 22 |
NEFEDOV V I, SALYN Y V, LEONHARDT G, et al. A comparison of different spectrometers and charge corrections used in X-ray photoelectron spectroscopy[J]. Journal of Electron Spectroscopy and Related Phenomena, 1977, 10(2):121-124.
doi: 10.1016/0368-2048(77)85010-X |
| 23 |
MORGAN W E, VAN WAZER J R. Binding energy shifts in the X-ray photoelectron spectra of a series of related group Ⅳ a compounds[J]. The Journal of Physical Chemistry, 1973, 77(7):964-969.
doi: 10.1021/j100626a023 |
| 24 | MORGAN W E, VAN WAZER J R, STEC W J. Inner-orbital photoelectron spectroscopy of the alkali metal halides,perchlorates,phosphates,and pyrophosphates[J]. Journal of the American Che- mical Society, 1973, 95(3):751-755. |
| 25 | JANSEN M, TRINSCHEK D. Phasenbeziehungen und natriumionenleitung im quasi-binären system Na2SiF6/Na3AlF6 [J]. Zeits-chrift für Anorganische Und Allgemeine Chemie, 1993, 619(1):88-92. |
| 26 |
GOL'DANSKII V I. The periodic law and progress in the study of the structure of matter[J]. Russian Chemical Reviews, 1974, 43(12):1021-1042.
doi: 10.1070/RC1974v043n12ABEH001879 |
| 27 |
LO P H, TSAI W T, LEE J T, et al. Role of phosphorus in the electrochemical behavior of electroless Ni-P alloys in 3.5 wt.% NaCl solutions[J]. Surface and Coatings Technology, 1994, 67(1/2):27-34.
doi: 10.1016/S0257-8972(05)80023-4 |
| [1] | YAN Xin,WEI Yilan. Present situation and prospect of comprehensive utilization of waste residue containing calcium and magnesium [J]. Inorganic Chemicals Industry, 2022, 54(1): 7-11. |
| [2] | ZHOU Jiaqi,CHEN Kui,WU Bin,JI Lijun,WU Yanyang. Study on kinetics of phosphate tailings calcination and acid leaching process [J]. Inorganic Chemicals Industry, 2022, 54(1): 77-82. |
| [3] | Zheng Jianguo,Yu Nanshu,Liu Yongxiu,Song Tao,Hu Zhaoping. Study on technological conditions of polymeric calcium magnesium phosphate fertilizer from phosphoric acid activated phosphate tailings [J]. Inorganic Chemicals Industry, 2020, 52(2): 43-46. |
| [4] | Zhang Pinghua,Yan Yunjie,Chen Jianjun,Wang Hongyan,Zhang Chunli,Wu Ning. Preparation of nanometer calcium carbonate from high-Ca-Mg phosphorus tailings by acid leaching [J]. Inorganic Chemicals Industry, 2019, 51(3): 63-66. |
| [5] | ZHAO Bo, CHEN Yan-Xin, YAO Yan-Fei. Research on suspension calcination of dolomitic phosphorus tailings [J]. INORGANICCHEMICALSINDUSTRY, 2013, 45(5): 18-. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||
|
||