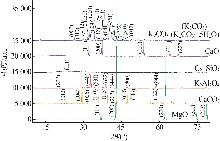
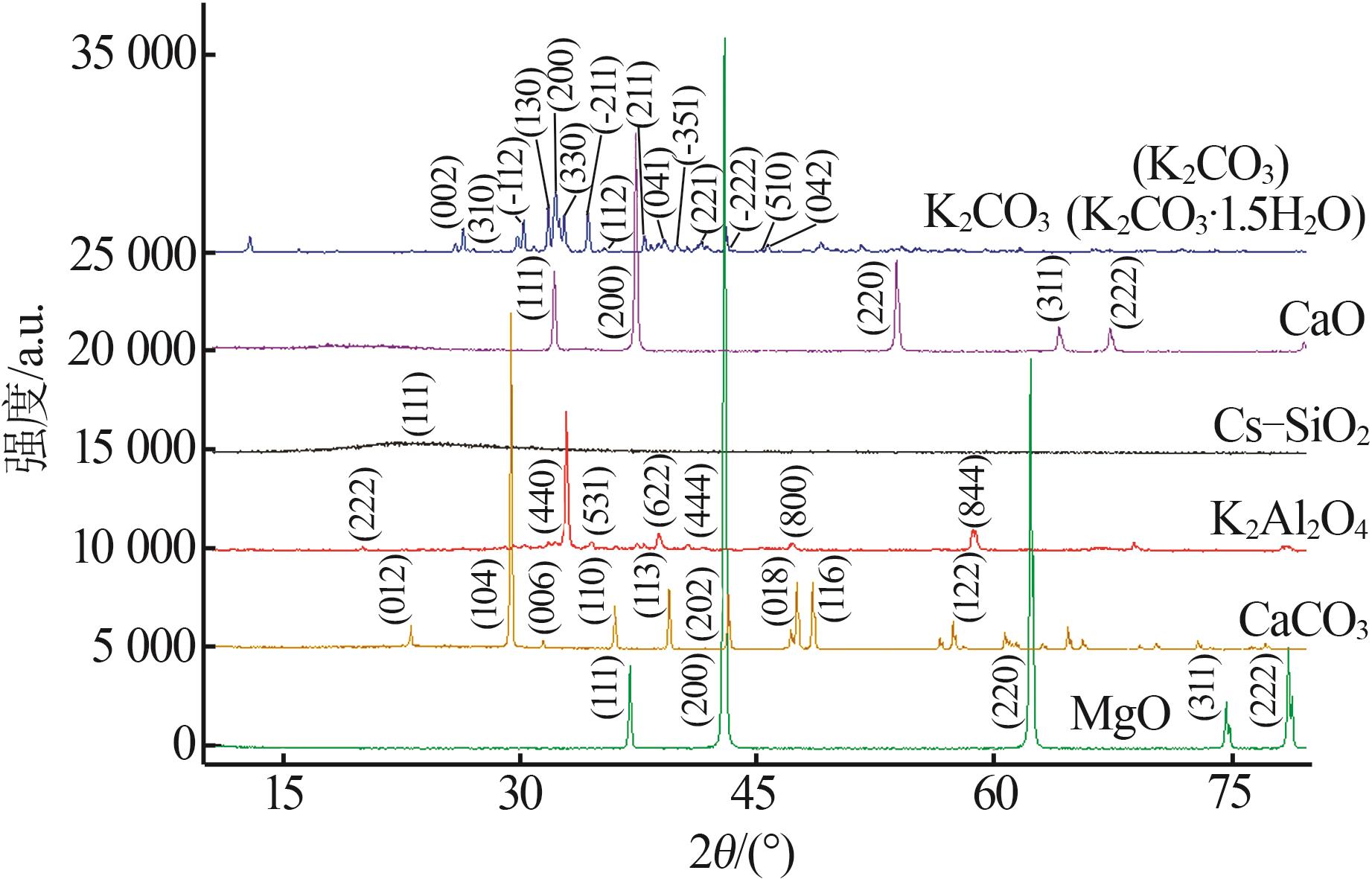
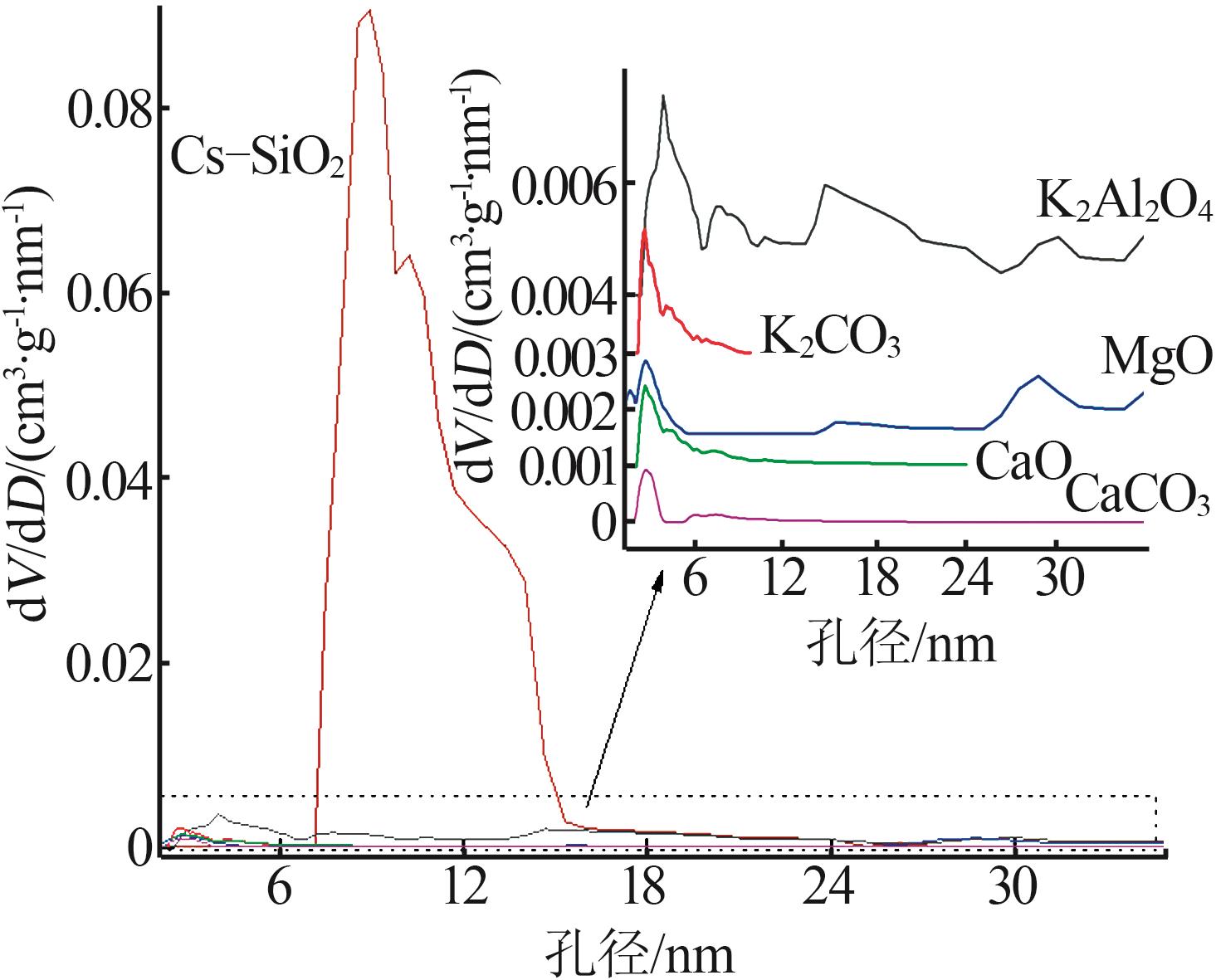
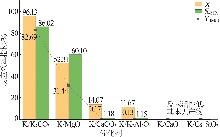
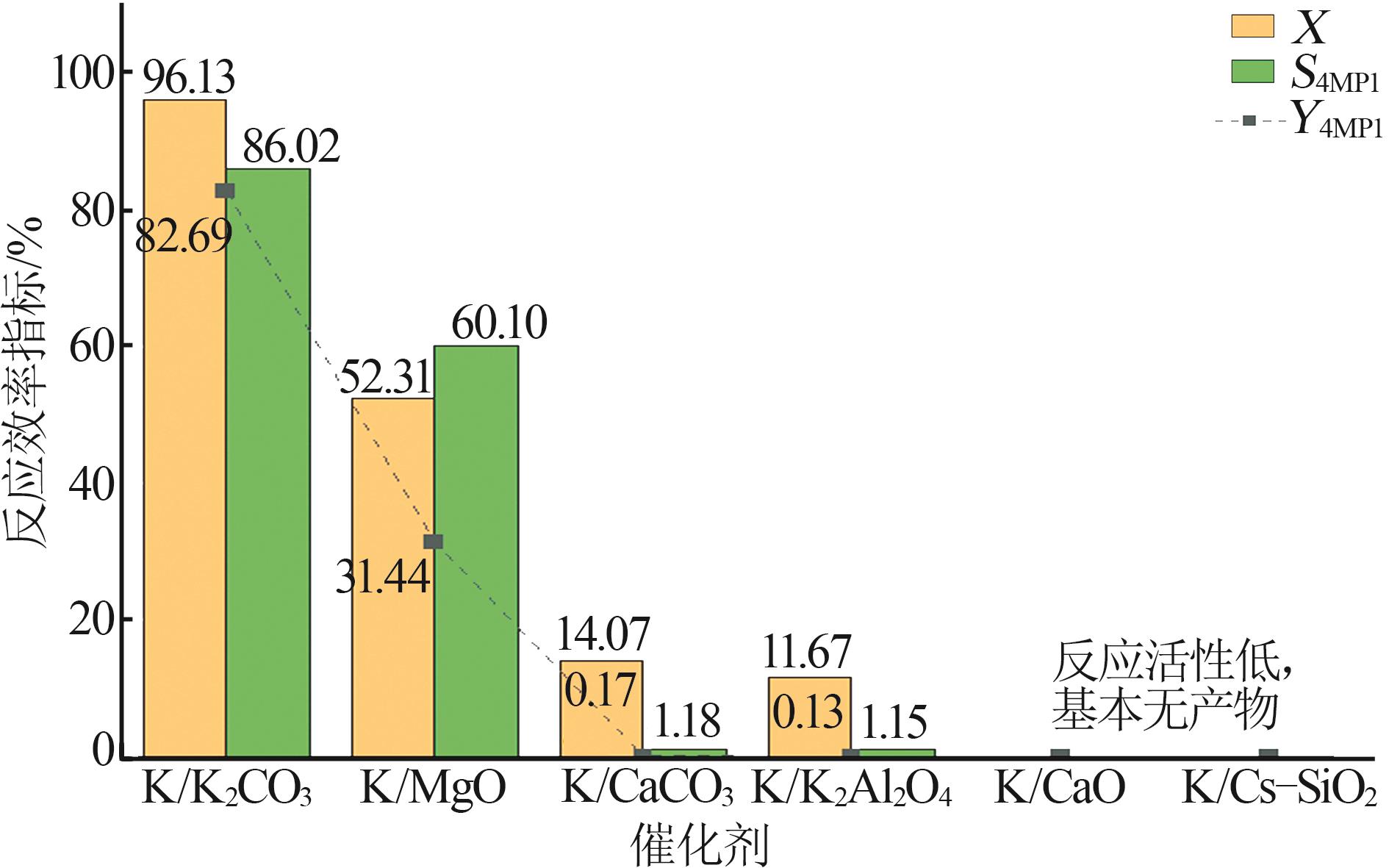
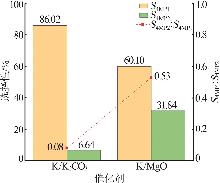
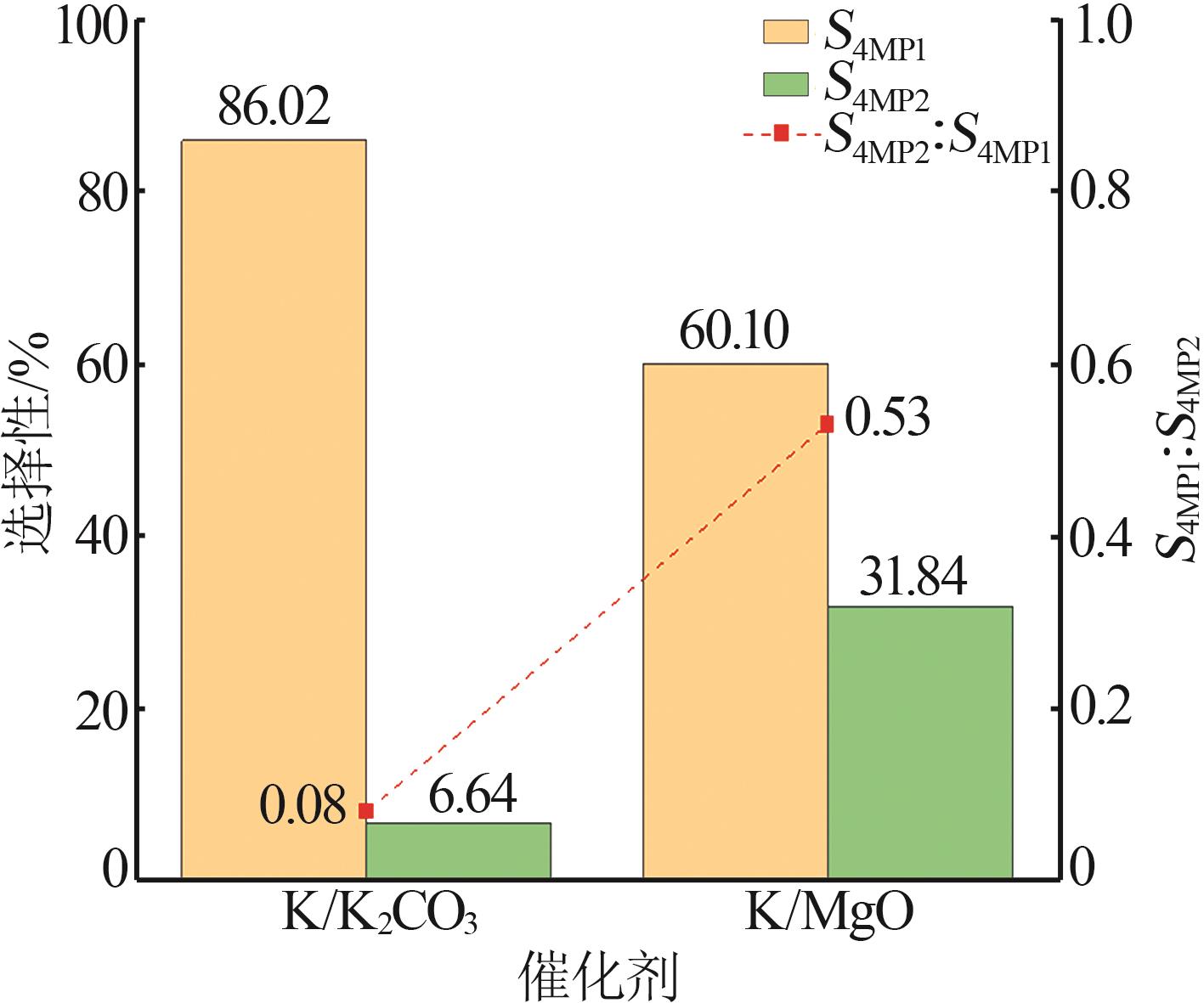
| [1] |
李文学,韩东,杨卫兰.中国丙烯及下游产品发展回顾及展望[J].现代化工,2024,44(5):11-14.
|
|
LI Wenxue, HAN Dong, YANG Weilan.Review and outlook on development of China′s propylene and its downstream products[J].Modern Chemical Industry,2024,44(5):11-14.
|
| [2] |
张垚,唐毓婧,郭子芳,等.聚4-甲基-1-戊烯的合成、结构与应用研究进展[J].合成树脂及塑料,2023,40(3):62-67,86.
|
|
ZHANG Yao, TANG Yujing, GUO Zifang,et al.Synthesis,structure and application of poly(4-methyl-1-pentene)[J].China Synthetic Resin and Plastics,2023,40(3):62-67,86.
|
| [3] |
高海洋,杨佳豪,涂广水,等.聚4-甲基-1-戊烯的结构、合成及应用[J].炼油与化工,2024,35(4):12-17.
|
|
GAO Haiyang, YANG Jiahao, TU Guangshui,et al.Structure,synthesis and application of poly4-methyl-1-pentene[J].Refining and Chemical Industry,2024,35(4):12-17.
|
| [4] |
秦龙,高彦山,唐勇.丙烯选择性齐聚合成重要化学品的研究进展[J].化学世界,2023,64(6):377-389.
|
|
QIN Long, GAO Yanshan, TANG Yong.Progress in synthesizing important chemical products via selective oligomerization[J].Chemical World,2023,64(6):377-389.
|
| [5] |
刘蕾,姚文君,谢元,等.丙烯齐聚合成低碳烯烃研究进展[J].炼油与化工,2024,35(4):18-23.
|
|
LIU Lei, YAO Wenjun, XIE Yuan,et al.Research progress on the polymerization of propylene into low carbon olefins[J].Refining and Chemical Industry,2024,35(4):18-23.
|
| [6] |
HASSELBRING L C. Product and process of hydrogen treatment of olefin dimerization catalysts:US,5081093A[P].1992-01-14.
|
| [7] |
IMAI H; MATSUNO M; KUDOH M.Process for preparing 4-methyl-1-pentene:EP,19820300065[P].1982-01-07.
|
| [8] |
KEIJI K, KEIGO K, TAKAHIRO A.Catalyst composition suitable for the dimerization or codimerization of alpha-olefins:EP,0083083A1[P].1983-07-06.
|
| [9] |
张明森,柯丽,杨菁,等.丙烯二聚合成4-甲基-1-戊烯[J].石油化工,2002,31(9):737-740.
|
|
ZHANG Mingsen, KE Li, YANG Jing,et al.Study on dimerization of propylene to produce 4-methyl-1-pentene[J].Petrochemical Technology,2002,31(9):737-740.
|
| [10] |
柴婧,靳海波,杨索和,等.超强碱催化剂载体结构对丙烯二聚生成4-甲基-1-戊烯反应性能影响的研究[J].过程工程学报,2023,23(6):918-924.
|
|
CHAI Jing, JIN Haibo, YANG Suohe,et al.Influence of support structure of superbase catalyst on dimerization of propylene to 4-methyl-1-pentene[J].The Chinese Journal of Process Engineering,2023,23(6):918-924.
|
| [11] |
仝洪凯.4-甲基-1-戊烯合成的固体碱催化剂及工艺设计[D].大连:大连理工大学,2021.
|
|
TONG Hongkai.Design of solid base catalysts and their technological process for propylene dimerization to produce 4-methyl-1-pentene[D].Dalian:Dalian University of Technology,2021.
|
| [12] |
孙洪伟,段庆华,郭战勇,等.丙烯二聚合成4-甲基戊烯-1的催化剂研究[J].精细石油化工,2003,20(1):14-17.
|
|
SUN Hongwei, DUAN Qinghua, GUO Zhanyong,et al.Study on the catalyst for synthesis of 4-methyl-1-pentene by propylene dimerization[J].Speciality Petrochemicals,2003,20(1):14- 17.
|
| [13] |
张伟,贾鑫,王兴永,等.催化剂酸碱性与催化酯醛缩合性能的关系[J].精细化工,2024,41(3):687-696.
|
|
ZHANG Wei, JIA Xin, WANG Xingyong,et al.Relationship between catalyst acid-base property and condensation performance for ester aldehydes[J].Fine Chemicals,2024,41(3):687-696.
|
| [14] |
王晓军,鲁妮,付源益,等.二氧化锆负载碱金属催化剂的制备及其催化葡萄糖转化性能[J].石油学报(石油加工),2022,38(5):1213-1219.
|
|
WANG Xiaojun, LU Ni, FU Yuanyi,et al.Preparation of zirconium dioxide supported alkali metal catalyst and its catalytic performance for glucose conversion[J].Acta Petrolei Sinica(Petroleum Processing Section),2022,38(5):1213-1219.
|
| [15] |
靳海波,王启维,江恒,等.一种固体超强碱催化剂的制备方法:中国,108554430A[P].2018-09-21.
|
| [16] |
王启维.碱金属负载型固体超强碱催化丙烯二聚反应研究[D].北京:北京石油化工学院,2015.
|
|
WANG Qiwei.Propylene dimerization on the alkali metal supported solid superbase[D].Beijing:Beijing Institute of Petrochemical Technology,2015.
|
| [17] |
张馨懿.原料杂质对丙烯聚合的影响分析[J].山西化工,2025,45(3):156-157.
|
|
ZHANG Xinyi.Analysis of the influence of raw material impurities on propylene polymerization[J].Shanxi Chemical Industry,2025,45(3):156-157.
|
| [18] |
赵永生.丙烯纯度对聚丙烯工艺的影响分析及调控对策[J].炼油与化工,2024,35(2):59-61.
|
|
ZHAO Yongsheng.Influence analysis of propylene purity on polypropylene process and control measures[J].Refining and Chemical Industry,2024,35(2):59-61.
|
| [19] |
JIN Haibo, CHAI Jing, YANG Suohe,et al.Catalytic reaction kinetics of propylene dimerization to 4-methyl-1-pentene using Cu-K/K2CO3 solid base catalyst[J].China Petroleum Processing & Petrochemical Technology,2024,26(1):78-87.
|
| [20] |
仝洪凯,陈霄,凌雨,等.适用于丙烯二聚反应非均相催化剂的研究进展[J].化工进展,2021,40(5):2546-2559.
|
|
TONG Hongkai, CHEN Xiao, LING Yu,et al.Advances in heterogeneous catalysts for propylene dimerization[J].Chemical Industry and Engineering Progress,2021,40(5):2546-2559.
|
| [21] |
江恒.合成4-甲基-1-戊烯的固体超强碱K/K2CO3催化性能研究[D].北京:北京化工大学,2017.
|
|
JIANG Heng.The catalytic characteristic of solid superbase K/K2CO3 in synthesis of 4-methyl-1-pentene[D].Beijing:Beijing University of Chemical Technology,2017.
|
| [22] |
江友法,刘程琳,罗孟杰,等.铝酸钾溶液自发分解过程中的氢氧化铝晶型转变[J].中国有色金属学报,2019,29(3):606-616.
|
|
JIANG Youfa, LIU Chenglin, LUO Mengjie,et al.Polymorphic transformation of aluminum hydroxide during spontaneous decomposition of supersaturated potassium aluminate solution[J].The Chinese Journal of Nonferrous Metals,2019,29(3):606- 616.
|
| [23] |
张昕,张萍波,范明明,等.K2O/MgFe2O4磁性固体碱催化剂的制备、表征及催化酯交换制备碳酸甘油酯[J].中国油脂,2024,49(5):71-77.
|
|
ZHANG Xin, ZHANG Pingbo, FAN Mingming,et al.Preparation,characterisation and catalytic transesterification for the preparation of glycerol carbonate of magnetic solid base catalyst K2O/MgFe2O4 [J].China Oils and Fats,2024,49(5):71-77.
|
| [24] |
时骏,张成喜,陈强.分子筛负载单原子金属催化剂的研究进展[J].石油学报(石油加工),2025,41(2):532-541.
|
|
SHI Jun, ZHANG Chengxi, CHEN Qiang.Research progress in zeolite supported single-atom metal catalysts[J].Acta Petrolei Sinica (Petroleum Processing Section),2025,41(2):532-541.
|
| [25] |
郑一帆.金属-载体相互作用在液-固多相催化反应中的影响[D].武汉:武汉大学,2022.
|
|
ZHENG Yifan.Evaluating the impact of metal-supportinteraction during liquid-solid heterogeneous catalysis[D].Wuhan:Wuhan University,2022.
|
| [26] |
蔡旭晖,林冠凯,王昊儒,等.LaNiO3- δ 薄膜中的氧缺位及其对金属输运行为的影响[J].低温物理学报,2022,44(1):33- 41.
|
|
CAI Xuhui, LIN Guankai, WANG Haoru,et al.Oxygen deficiency and its effect on the metallic transport behavior in LaNiO3- δ films[J].Low Temperature Physical Letters,2022,44(1):33-41.
|
| [27] |
王雨恒.负载型Na2SiO3的制备及其在酯交换合成DMC中的应用[D].重庆:重庆理工大学,2024.
|
|
WANG Yuheng.Preparation of supported Na2SiO3 and its application in transesterification synthesis of DMC[D].Chongqing:Chongqing University of Technology,2024.
|
| [28] |
朱远平.不对称烯烃的亲电加成反应的教学:运用软硬酸碱原理讨论马氏规则[J].科技创新导报,2008,5(1):235.
|
|
ZHU Yuanping.Teaching of electrophilic addition reaction of asymmetric olefins:Discussion on Mahalanobis rule by using the principle of soft and hard acids and bases[J].Science and Technology Innovation Herald,2008,5(1):235.
|
 ), HUA Jixiang1,2, ZHANG Huan1,2, ZHANG Shenghong1,2, SUN Peiyong1,2, YAO Zhilong1,2(
), HUA Jixiang1,2, ZHANG Huan1,2, ZHANG Shenghong1,2, SUN Peiyong1,2, YAO Zhilong1,2( )
)